


First whiteflies from the Eocene amber of Denmark
JOWITA DROHOJOWSKA, JOANNA ŚLADOWSKA, and JACEK SZWEDO
Drohojowska, J., Śladowska, J., and Szwedo, J. 2024. First whiteflies from the Eocene amber of Denmark. Acta Palaeontologica Polonica 69 (4): 649–676.
One new genus Gregorites Drohojowska & Szwedo with seven new species of whiteflies (Aleyrodidae) from Eocene amber collected in Denmark are described. All new taxa: Gregorites aegiri, G. bergelmiri, G. skadii, G. bestlae, G. thrymi, G. halogii, and G. ymiri are represents of the subfamily Aleurodicinae. The newly described genus appeared as highly diverse, with 7 new species, while remaining Paleogene Aleurodicinae are monospecific in vast majority. The key for identification of species of Aleurodicinae from Eocene Danish amber is provided. The morphological features of newly established taxa are discussed. A brief overview of the Aleyrodidae preserved in the Eocene fossil resins is presented, their taxonomic diversity and morphological disparity offered by the fossilised imagines are discussed. The importance of the fossil for phylogenetic, evolutionary and ecological studies is presented. Various factors influencing diversification and disparification of whiteflies at local and regional scales are discussed. The potential biases associated with the collection and preservation of specimens are briefly overviewed. Records of Aleyrodidae in the fossil resins of Eocene of Europe are discussed.
Key words: Hemiptera, Aleyrodoidea, Eocene, Danish amber, new taxa.
Jowita Drohojowska [jowita.drohojowska@us.edu.pl; ORCID: https://orcid.org/0000-0002-7668-2242 ] and Joanna Śladowska [joanna.j.sladowska@gmail.com; ORCID: https://orcid.org/0000-0002-3669-3430 ], Institute of Biology, Biotechnology and Environmental Protection, University of Silesia, 9, Bankowa St., PL40-007 Katowice, Poland.
Jacek Szwedo [jacek.szwedo@ug.edu.pl; ORCID: https://orcid.org/0000-0002-2796-9538 ] (corresponding author), Laboratory of Evolutionary Entomology and Museum of Amber Inclusions, Department of Invertebrate Zoology and Parasitology, University of Gdańsk, 59, Wita Stwosza St., PL80-308 Gdańsk, Poland.
Received 12 June 2024, accepted 15 October 2024, published online 19 December 2024.
Copyright © 2024 J. Drohojowska et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The Hemiptera is the largest monophyletic order of the hemimetabolous insects, one of the Big Five insect orders, characterised as having specialized piercing-sucking mouthparts—the rostrum, i.e., a bunch of penetrating mandibulary and maxillary stylets covered by a segmented labium (Grimaldi and Engel 2005; Szwedo 2018). The order is divided into six suborders: extinct Paleorrhyncha, and extant Sternorrhyncha, Fulgoromorpha, Cicadomorpha, Coleorrhyncha, and Heteroptera. Hemipteran insects are opophagous, feeding on plant phloem, xylem, and cell content, some heteropterans are predatory or haematophagous (Henry 2017; Bartlett et al. 2018; Szwedo 2018). The Sternorrhyncha, comprising approximately 19,000 described Recent species, represents a significant group of insects with both ecological and economic importance. This includes four major groups—aphids, phyloxerans and adelgids (Aphidomorpha), scale insects (Coccidomorpha), whiteflies (Aleyrodomorpha) and psyllids (Psylloidea). The fossil record of sternorrhynchans can be traced back to the Permian, but their evolutionary history goes back to the Carboniferous (Drohojowska et al. 2020), and the early stages of their evolution and diversification are poorly understood. Three extinct infraorders are known—Pincombeomorpha and Naibiomorpha—which are variously placed in classifications and relationships hypotheses, and Dinglomorpha—which is sister to Aleyrodomorpha. The placement of extinct Protopsyllidioidea is debatable (Drohojowska et al. 2020; Hakim et al. 2021). The prevailing view is that the Sternorrhyncha constitute a monophyletic lineage. Their sedentary lifestyles coupled with phloem-feeding behaviors in these insects, which are plant parasites, have driven morphological reductions and losses, neotenous females, extreme sexual dimorphism, and convergently derived morphological characters that would otherwise be useful in phylogenetic analyses. Consequently, reconstructing the relationships of Sternorrhyncha is rather challenging endeavour.
The Aleyrodidae, the sternorrhynchan hemipterans are commonly referred to as whiteflies. This name is derived from the presence of a powdery secretion, which covers their body and wings of the adults of almost all species. Similarly to other sternorrhynchans such as phyloxerans, adelgids, aphids, psyllids, and scale insects whiteflies use plant sap as their diet. Furthermore, most of them are closely related with particular host-plants (Gerling 1990; Grimaldi and Engel 2005; Evans et al. in press). The whiteflies (Aleyrodomorpha Chou, 1963) comprise a single family—Aleyrodidae Westwood, 1840, currently subdivided into four subfamilies: extinct Bernaeinae Shcherbakov, 2000, and Recent Aleyrodinae Westwood, 1840, Aleurodicinae Quaintance & Baker, 1913, and Udamoselinae Enderlein, 1909, the latter of which is of questionable taxonomic status (Evans et al. in press). If the numbers of described species are an accurate guide, then whiteflies are by far the least speciose of the four major extant groups of sternorrhynchan Hemiptera, with around 1700 currently valid Recent species (Evans et al. in press). The fossil record of the family Aleyrodidae extends to the Late Jurassic (Shcherbakov 2000; Drohojowska et al. 2019). Other fossils (but only a few formally described) were reported from the sedimentary deposits and fossil resins aged as Early Cretaceous, Late Cretaceous, Paleogene, and Neogene (Schlee 1970; Rietschel 1983; Poinar 1992; Shcherbakov 2000; Schmidt et al. 2010; Drohojowska and Szwedo 2011a, b, 2013a, b, 2015; Drohojowska et al. 2015, 2019, 2022; Szwedo et al. 2019). The occurrence of whiteflies in a fossilised state is relatively uncommon, and oftentimes only sole specimens are found.
Institutional abbreviations.—NHMD, Natural History Museum of Denmark, Copenhagen, Denmark; ZMUC, Zoological Museum of University of Copenhagen, Denmark.
Nomenclatural acts.—This published work and the nomenclatural acts it contains have been registered in ZooBank: urn:lsid:zoobank.org:pub:18F93AAA-39B3-479F-8814-1DDA82DE9691.
Geological setting
The Eocene fossil resins of Europe comprise the lowermost Eocene amber from Oise (Paris Basin, France) and younger resins collectively named “Baltic amber”, are aged variously (Lutetian to Priabonian). Recent studies suggests that the broad concept of “Baltic amber” in respect to inclusions, as Eocene succinite and accessory resins found in the Baltic Sea Basin (e.g., Rovno amber, Bitterfeld amber, etc.) should be abandoned, in favor of the more restrictive and precise definition of Baltic amber as the secondary amber accumulation found only in the Gulf of Gdańsk (Sambia Peninsula to Chłapowo). Deposits of amber in Ukraine (Rovno-Zhitomir region), amber from Bitterfeld (Lower Saxony, Germany), amber from Górka Lubartowska (Lublin region, Poland) were probably alimented from independent geographical sources (Wolfe et al. 2009, 2016; Mitov et al. 2021; Radchenko et al. 2021; Słodkowska et al. 2022); the same reservation seems to be true for amber deposits found on Denmark coast (Larsson 1978). The provenance of the amberiferous deposits from which the Danish coast amber could have been washed out is currently unknown. The inclusions in the amber described below are most likely derived from the west coast of Jutland. According to Larsson (1978: 33), coastal amber was found in “good amounts from Fanø to Nissum Bredning”. However, the majority of the amber was collected on the stretch between Blåvandshuk and Hvidesande. Larsson (1978) proposed that Danish amber was redeposited to Miocene lignites of Jutland from South Swedish Eocene forests, which is an area different from the hypothesised areas of origin for the amber deposit of the Gulf of Gdańsk. This idea is consistent with more recent models of the evolution of the North Sea (Gibbard and Lewin 2016; Dybkjær et al. 2020). Currently, amber is collected on the southwestern shores of Denmark, and on the shores of the island of Fanø (Simonsen 2008; Klug and Klug 2015). It should be noted that fossilised resin of probable araucarian origin was found in older layers of the Fur Formation (Pedersen and Pedersen 2013).
Material and methods
The majority of the specimens described in this work were previously studied in the late 1960s by Schlee (1970). He presented only general morphology and measurements of thirteen specimens (five males, four females, and four of unknown sex), due to difficulties in interpreting the imagines of the whiteflies, their morphology, a lack of comparative studies and material, and the poor condition of the specimens (Schlee 1970). The specimens were from NHMD (all specimens examined here with additional ZMUC acquisition numbers) and all were inclusions from the Eocene amber were collected in Denmark (so-called Danish, or Scandinavian amber). All specimens are inclusions in tiny pieces of amber devoid of any syninclusions. For the microscopic examination, we used a Nikon SMZ1500, Nikon SMZ1270, Nikon SMZ25, Leica M205C stereoscopic microscopes and a Nikon Microphot-FX equipped with a camera Lucida and interchangeable direct and transmitted light. The photographs were taken using a Nikon Microphot-FX with a Nikon Eclipse E 600 digital camera and Lucia® software and then edited with Adobe® Photoshop Elements 6.0.
The general morphological terminology adheres to Weber (1935); the terminology of the thoracic sclerites is according to Drohojowska and Szwedo (2011a); the venation nomenclature follows the suggestions of Shcherbakov (2000) modified by Drohojowska and Szwedo (2013a), and the measurements are in accordance with the model proposed by Martin (2007).
Systematic palaeontology
Class Insecta Linnaeus, 1758
Order Hemiptera Linnaeus, 1758
Suborder Sternorrhyncha Amyot & Audinet-Serville, 1843
Infraorder Aleyrodomorpha Chou, 1963
Family Aleyrodidae Westwood, 1840
Subfamily Aleurodicinae Quaintance & Baker, 1913
Key to genera of succinite Aleyrodidae
1. Fore wing with a single, straight, non-forked central vein (Rs present, R1 usually short or absent); females with 2 pairs of ventral abdominal plates and males with 4 pairs of ventral abdominal wax plates; tarsal paronychium usually thickened and clothed with spinules (microsetae) on the plantar surface ................................................................................. subfamily Aleyrodinae (Snotra)
– Fore wing with a forked, central vein (Rs present, R1 strongly developed, except Rovnodicus, which has R1 short); females with 4 pairs of ventral abdominal plates and males with 3 pairs of ventral abdominal wax plates; tarsal paronychium usually thin and spine-like subfamily Aleurodicinae (Paernis, Rovnodicus, Gregorites Drohojowska & Szwedo gen. nov., Medocellodes) ................................................................................................................. 2
2. CuA on fore wing long and well visible, antennae seven-segmented, all antennomeres separated ................................................................ 3
– CuA on fore wing not visible, antennae six- or seven-segmented (then only segments 1st to 5th separated, remaining segments fused, in the form of narrow flagellum, longer than half the combined length of the preceding segments) ........................................................ 4
3. Flagellum acute, median ocellus absent; amber from Denmark ....................................... Gregorites Drohojowska & Szwedo gen. nov.
– Flagellum not acute, with two short terminal setae; median ocellus present; amber from Gulf of Gdańsk ................................................................................... Medocellodes Drohojowska & Szwedo, 2022
4. Antenna seven-segmented, fore wing broad, 2.1 times as long as wide; branch R1 on fore wing well visible, long and shallowly curved, directed anteriad; branch Rs straight, directed towards posteroapical angle of fore wing; hind wing with R vein forked; amber from Gulf of Gdańsk ......... Paernis Drohojowska & Szwedo, 2011
– Antenna six-segmented, fore wing narrow, 2.9 times as long as wide; branch R1 on fore wing weakly visible, short and straight; branch Rs straight at base, directed towards middle of apical margin, slightly arcuate in apical portion; hind wing with R vein not forked; amber from Ukraine ........ Rovnodicus Drohojowska & Szwedo, 2015
Genus Gregorites Drohojowska & Szwedo nov.
ZooBank LSID: urn:lsid:zoobank.org:act:0F109DE4-A009-4008-8D7 B-D054C949870A.
Etymology: Dedicated to the son of JD—Grzegorz (in Latin Gregorius). Gender: masculine.
Type species: Gregorites aegiri Drohojowska & Szwedo sp. nov., by present designation.
Diagnosis.—Antennae seven-segmented (as in Paernis Drohojowska & Szwedo, 2011), all antennomeres separated, flagellum short and acute (in Paernis only antennomeres 1–5 separated, remaining antennomeres fused, in the form of a narrow flagellum, longer than half the combined length of the preceding antennomeres), 3rd antennomere the longest. Fore wing widening apicad, the widest in about 2/3 of the length. Branch R1 well developed, vein CuA on fore wing long and well visible (not visible in Rovnodicus Drohojowska et Szwedo, 2015, and Paernis).
Stratigraphic and geographic range.—Eocene (Lutetian–Priabonian); western coast of Denmark.
Gregorites aegiri Drohojowska & Szwedo sp. nov.
Fig. 1.
ZooBank LSID: urn:lsid:zoobank.org:act:D5415248-79AE-4039-92E6- 401B9054DFFD.
Etymology: Specific name is after Ægir, a giant in Norse mythology, ruler of the seas.
Holotype: Male, NHMD 38959 [alternative ZMUC 902132], collected by C.V. Henningsen, 9.3.1967, deposited in Amber Collection in Natural History Museum of Denmark. A well preserved and complete imago of whitefly is included in clear yellowish and very small sized piece of amber (5×4×3 mm) in shape of a flat cube.
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago, male. Antenna seven-segmented, scapus wider than basal part of pedicel, 4th antennomere longer than 5th, antennomeres 6th, and 7th the same length. Lateral part of pronotum triangular, mesopraescutum with rounded lateral angles, slightly wider than length of mesopraescutum in midline. Mesoscutum almost the same length as pronotum, posterior margin with gentle, arcuate depression, lateral part strongly elongated. Anterior margin of mesoscutellum in middle part rounded, in lateral part directed posteriad. Mesopostnotum with gently rounded anterior margin, slightly wider than its length in midline, anterior angles rounded, posterior angle wide arched. Anterior margins of metascutum rounded, posterolateral margins visibly rounded, posterior margin strongly rounded. Anterior margin of metascutellum in the middle part strongly rounded. Fore wing with vein CuA present, branch Rs slightly longer than branch R1, claval vein CuP much longer than half of length of fore wing, branch Sc+R in hind wing not reaching margin. Claspers strongly elongated, inner margin with depression near base of aedeagus. Aedeagus funnel-like shape, bulbous basally, narrowed and straight distally.
Thorax well visible on dorsal side (Fig. 1A4, A9, A11). Pronotum slightly wider than head with compound eyes, similar width as mesoscutum. Lateral part of pronotum triangular, longer than length of pronotum in midline, slightly curved anteriad. Anterior and posterior margins of pronotum arched. Mesopraescutum triangular and clearly separated, slightly wider than length in midline. Mesopraescutum 2.4 times narrower than pronotum. Anterior margin arched, lateral angles rounded, posterolateral margins straight, converging at an angle of 90°. Mesoscutum M-shaped, narrower in middle part with rounded anterior angles, 9 times wider than length in midline. Lateral part in the form of relatively long, narrow and blunt convexity. Anterolateral and posterolateral margins with clear depressions. Posterior margin concave in the middle. Mesoscutellum narrow, band-like, with the middle part clearly wider and rounded. Anterior margin in the middle rounded, posterior margin almost straight. Lateral parts of mesoscutellum narrow and slightly directed posteriad. Mesopostnotum triangular, with gently rounded anterior margin, slightly wider than long in midline; anterior angles rounded, posterolateral margins arched, posterior angle wide and arched, visible incision in midline. Metascutum partly covered by wings, in the middle part visibly narrowed, several times wider than the length in the midline. Incision in the middle part widely arcuate, anterior margin rounded, the lateral part in the form of narrow and blunt protuberances directed anteriad, the posterolateral margins rounded, the posterior margin is strongly rounded. Metascutellum narrow laterally, clearly wider in the middle, anterior margin rounded, posterior margin almost straight in the middle, slightly arched in the posterior-lateral parts; narrow lateral bands directed anteriad. Parapteron relatively large and oval (Fig. 1A4, A9), tegula subtriangular (Fig. 1A4, A9).
Fore wing slightly concave in the middle part (Fig. 1A1, A8, A10, A11), widening apicad, the widest in 1/3 of the length, about 2 times as long as wide. Costal vein strongly curved, anteroapical angle broadly rounded, apical margin and posteroapical angle rounded. Posterior margin rounded basally, further almost straight. Anterior margin covered with very small tubercle and setae (Fig. 1A10, A11). Common stem Sc+R+CuA short and thick. Branch Sc+R straight, directed towards the anteroapical angle, forked before half the length of the fore wing. Branch R1 straight, not reaching to the anterior margin. Branch Rs straight, about 2.1 times longer than the branch R1, not reaching to the anterior margin (Fig. 1A1, A11). Vein CuA straight, slightly curved in the middle part, departing at an acute angle from the common branch Sc+R+CuA, not reaching to the anterior margin (Fig. 1A1, A10, A11). Claval vein CuP straight, slightly curved in the middle part, reaching over half of fore wing length, not reaching to fore wing margin (Fig. 1A1, A11). Hind wing relatively broad basally, widening apicad, widest in 2/3 of the hind wing length, about 2.2 times as long as wide (Fig. 1A2, A8, A10, A11). Anterior margin of hind wing invisible, partly covered by the fore wing and partly rolled up, angle and apical margin rounded, posteroapical angle wide rounded. Posterior margin of hind wing rounded. Branch Sc+R slightly curved in the middle, not reaching margin.
Description.—Measurements: Table 1. Head on ventral side covered with mold (Fig. 1A8, A12). Compound eyes relatively large, bulging and not divided (Fig. 1A9, A11). Ommatidia similar size. Lateral ocelli placed in front and above compound eyes, visible on dorsal side (Fig. 1A9). Anterior margin on vertex slightly rounded without median incision (Fig. 1A9), vertex with coronal suture distinct (Fig. 1A9). Antennae seven-segmented (Fig. 1A6, A9, A13). Scapus more twice as wide as long, pedicel large and massive, wider in apical part, about 2.3 times longer than width. Third antennomere the longest, about twice longer than antennomere 4th. Fourth antennomere 1.6 times longer than 5th. Antennomeres 6th and 7th the same length. Seventh antennomere tapering apicad.
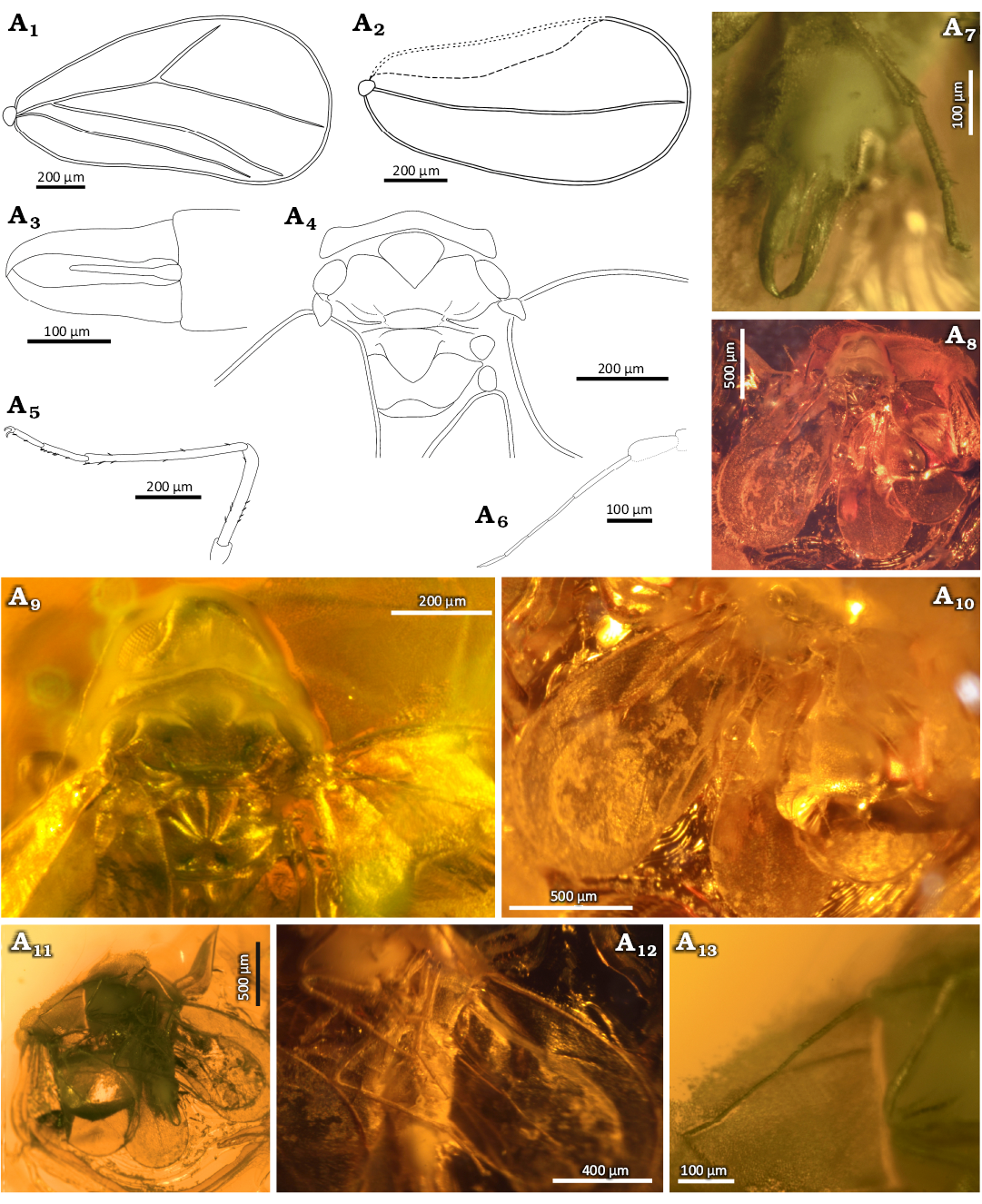
Fig. 1. Whitefly Gregorites aegiri Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38959 / ZMUC 902132), Eocene, Danish amber. A1, fore wing; A2, hind wing; A3, genitalia; A4, thorax; A5, metaleg; A6, A13, antenna; A7, genitalia and metatarsus; body in ventral (A8) and dorsal (A11) views; A9, head and thorax; A10, wings; A12, legs. Explanatory drawings (A1–A6), photographs (A7–A13).
Proleg the shortest, metaleg the longest (Fig. 1A5, A8, A12). Coxae elongated, slightly conical. Trochanters slightly longer than their width. Femora shorter than tibiae. Tarsus two-segmented. Profemur not visible, covered with milky veil. Metatibia 1.9 times longer than metafemur. basimetatarsomere 1.3 times longer than apical metatarsomere. Metatarsus similar length as metafemur. Hooked claw visible in metaleg (Fig. 1A4, A7), paronychium invisible. Metafemur and metatibiae with few, rare and scarcely distributed setae (Fig. 1A5, A7, A8, A12). Singular setae on metatarsus, the most in the apical part of basimetatarsomere (Fig. 1A5, A7).
The abdomen from dorsal side covered by wings (Fig. 1A10, A11), from the ventral side slender and tapering towards apex (Fig. 1A8, A12). Abdomen with genitalia longer than the total length of the head and thorax (Fig. 1A3, A7, A8). Wax plates invisible. Pregenital segments obscured by mold (Fig. 1A7, A8, A12). Aedeagus relatively long, thin and straight, wider (funnel-shaped) basally (Fig. 1A3, A7), its length exceeds half the length of claspers. Claspers placed laterally, elongated, wider basally, tapered in apical half (Fig. 1A3, A7). The inner margin of claspers is slightly arched, with cavities near the base of the aedeagus, further straight and curved at the apex. The tops of claspers are pointed, overlapping, with a few small setae on the outside.
Stratigraphic and geographic range.—Type locality and horizon only.
Gregorites bergelmiri Drohojowska & Szwedo sp. nov.
Fig. 2.
ZooBank LSID: urn:lsid:zoobank.org:act:429FC82E-9E21-42E7-AD 00-F68B5F435CD8.
Etymology: After Bergelmir, in the Norse mythology the giant who survived the flood of Ymir blood, ancestor of new line of giants.
Holotype: Male, NHMD 38972 [alternative ZMUC 902145], collected by A.K. Andersen, 28.3.1968, deposited in Amber Collection in Natural History Museum of Denmark. A well preserved and complete imago of whitefly is included in clear yellowish and moderate sized piece of amber (28×16 mm) of irregular shape.
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago, male. Antenna with 4th and 5th antennomeres the same length, 4th and 5th antennomeres shorter than 6th antennomere. Pronotum as wide as mesoscutum, lateral part triangular, much longer than their length line. Mesopraescutum small, poorly marked and asymmetrical, anterior margin curved, posterolateral margins gently, posterior angle widely arched. Anterior margin of mesoscutum widely arched, lateral margins strongly elongated and concave, posterolateral parts relatively large, tapering apically convexity, posterior margin strongly rounded. Mesoscutellum narrow and strongly concave anteriad. Mesopostnotum diamond-shaped, anterior margin strongly rounded. Metascutum with deeply, arcuately incised in median portion, posterior margin slightly rounded in the lateral part and almost straight in median portion. The middle part of metascutellum lenticular shape. Fore wing with vein CuA, branch Rs 2 times longer than branch R1. Hind wing short and very wide, with branch Rs 1.8 times as long as branch R1. Pygofer wider than its length in the middle line. Claspers about 1.3 times longer than the pygofer, with wavy outer and inner margins.
Description.—Measurements: Table 1. Compound eyes relatively large, not bulging and not divided (Fig. 2A9, A11, A12). Ommatidia small and equal in size. Vertex with coronal suture distinct (Fig. 2A9, A11). Anterior margin of vertex slightly concave in the middle part, but not indented (Fig. 2A9, A11, A12). Rostrum (Fig. 2A12) short and massive, reaching mesocoxa. Lateral ocelli relatively small located above compound eyes (Fig. 2A12).
Table 1. Basic measurements of the specimens of Gregorites Drohojowska & Szwedo gen. nov. species from Danish amber (in mm). Measurements of thorax tergites only in midline. * composite length of antennomeres 3rd to 7th; ** only anterior and posteriori margin of wax plates complex are visible.
|
|
G. aegiri |
G. |
G. |
G. |
G. |
G. |
G. |
902143 |
902144 |
902146 |
902136 |
|
Body length total (including claspers) |
1.34 |
1.30 |
1.08 |
1.07 |
1.32 |
1.39 |
– |
0.90 |
1.23 |
0.87 |
0.81 |
|
Head length in midline |
0.12 |
0.13 |
– |
– |
– |
0.23 |
0.13 |
0.16 |
– |
– |
– |
|
Head with compound eyes width |
0.31 |
0.32 |
– |
– |
– |
0.32 |
– |
0.21 |
– |
– |
– |
|
Scapus length |
0.04 |
0.03 |
– |
– |
– |
0.01 |
– |
0.02 |
– |
– |
– |
|
Pedicel length |
0.1 |
0.08 |
– |
0.06 |
0.05 |
0.06 |
0.08 |
0.05 |
0.07 |
– |
0.06 |
|
3rd antennomere |
0.16 |
0.16 |
0.28* |
0.15 |
0.15 |
0.17 |
0.13 |
0.12 |
0.16 |
– |
– |
|
4th antennomere |
0.08 |
0.06 |
0.05 |
0.07 |
0.11 |
0.06 |
0.03 |
0.04 |
– |
– |
|
|
5th antennomere |
0.05 |
0.06 |
0.06 |
0.07 |
0.09 |
– |
0.04 |
0.05 |
– |
– |
|
|
6th antennomere |
0.06 |
0.07 |
0.05 |
0.06 |
0.04 |
– |
0.03 |
0.04 |
– |
0.04 |
|
|
7th antennomere |
0.06 |
0.04 |
0.04 |
0.06 |
0.06 |
– |
0.04 |
0.05 |
– |
0.03 |
|
|
Fore wing length |
1.37 |
1.41 |
1.38 |
1.21 |
1.26 |
1.62 |
1.36 |
0.95 |
1.32 |
1.01 |
0.93 |
|
Fore wing width |
0.70 |
0.60 |
0.56 |
0.51 |
0.56 |
0.84 |
0.58 |
0.42 |
0.61 |
0.36 |
0.48 |
|
Hind wing length |
1.08 |
1.01 |
1.10 |
0.96 |
1.11 |
– |
1.21 |
0.83 |
1.07 |
0.76 |
0.93 |
|
Hind wing width |
0.50 |
0.58 |
0.48 |
0.36 |
0.47 |
– |
0.46 |
0.34 |
– |
0.31 |
0.33 |
|
Profemur length |
– |
|
– |
– |
0.16 |
0.28 |
– |
0.13 |
– |
– |
0.14 |
|
Protibia length |
0.29 |
0.29 |
0.25 |
– |
0.25 |
0.35 |
– |
0.20* |
– |
– |
0.16 |
|
Probasitarsomere length |
0.12 |
0.12 |
0.11 |
– |
0.10 |
0.17 |
– |
0.09 |
– |
– |
0.08 |
|
Proapicaltarsomere length |
0.09 |
0.09 |
0.08 |
0.09 |
0.08 |
0.10 |
– |
0.06 |
– |
– |
0.06 |
|
Length of claw proleg |
0.01 |
– |
0.01 |
0.01 |
0.02 |
0.01 |
– |
0.01 |
– |
– |
0.01 |
|
Pronotum width |
0.36 |
0.37 |
0.31 |
– |
– |
– |
– |
– |
– |
– |
– |
|
Mesopraescutum length |
0.13 |
0.08 |
0.1 |
– |
– |
– |
0.12 |
– |
– |
– |
– |
|
Mesopraescutum width |
0.15 |
0.17 |
0.14 |
– |
– |
– |
0.17 |
– |
– |
– |
– |
|
Mesoscutum length |
0.05 |
0.02 |
0.06 |
– |
– |
– |
0.05 |
– |
– |
– |
– |
|
Mesoscutum width |
0.37 |
0.37 |
0.30 |
– |
– |
– |
0.36 |
– |
– |
– |
– |
|
Mesoscutellum length |
0.04 |
0.02 |
0.03 |
– |
– |
– |
0.05 |
– |
– |
– |
– |
|
Mesoscutellum width |
0.15 |
– |
0.13 |
– |
– |
– |
0.17 |
– |
– |
– |
– |
|
Mesopostnotum length |
0.11 |
0.15 |
0.08 |
0.12 |
– |
– |
0.10 |
– |
– |
– |
– |
|
Mesopostnotum width |
0.13 |
0.12 |
0.08 |
0.12 |
– |
– |
0.13 |
– |
– |
– |
– |
|
Metascutum length |
0.04 |
0.01 |
0.03 |
0.04 |
– |
– |
0.03 |
– |
– |
– |
– |
|
Metascutum width |
– |
0.27 |
0.23 |
– |
– |
– |
– |
– |
– |
– |
– |
|
Metascutellum length |
0.05 |
0.02 |
0.03 |
0.03 |
– |
– |
0.04 |
0.03 |
– |
– |
– |
|
Metascutellum width |
0.12 |
0.11 |
0.15 |
0.12 |
– |
– |
0.11 |
0.10 |
– |
|
– |
|
Abdomen length (with claspers) |
0.76 |
0.72 |
0.56 |
0.54 |
0.76 |
0.76 |
– |
0.49 |
0.69 |
|
0.42 |
|
Wax plate I |
– |
0.26** |
– |
? |
– |
|
|
|
|
|
0.07 |
|
Wax plate II |
– |
– |
0.10 |
– |
0.09 |
|
|
|
|
0.06 |
|
|
Wax plate III |
– |
– |
0.11 |
– |
0.08 |
|
|
|
|
0.05 |
|
|
Wax plate IV |
– |
– |
|
– |
|
|
|
|
|
0.05 |
|
|
Claspers length |
0.21 |
0.21 |
– |
– |
0.19 |
0.16 |
– |
0.11 |
– |
– |
– |
Antennae seven-segmented (Fig. 2A3, A8, A11, A12). Scapus weakly visible, slightly wider than its length, pedicel large and massive, wider apicad, about 1.7 times as long as wide. Third antennomere the longest, about 3.2 times longer than 4th one. Antennomeres 4th and 5th the same length, 6th one 1.2 times longer than antennomeres 4th and 5th. Seventh antennomere about 1.5 times shorter than 6th, tapering towards apex.
Thorax well visible (Fig. 2A6, A9, A11). Pronotum slightly wider than the head with compound eyes, about as wide as mesoscutum, longer at sides. Anterior and posterior margins triangular shape. Pronotum with lateral protruding parts, with rounded angles anteriad. Mesopraescutum small, poorly marked and asymmetrical, anterior margin strongly arched, posterolateral margins slightly arcuate, converging in the middle, wider than its midline length, posterior angle widely arcuate. Mesoscutum M-shaped, with a wide arcuate deep indentation at the anterior margin, clearly wider than its length in the midline. Anterolateral margins rounded, lateral margins strongly elongated and concave. Posterolateral parts of mesoscutum in the form of relatively large, tapering apically bulges, posterior margin rounded. Mesoscutellum in the form of a long, narrow and strongly curved transverse plate, towards anteriad. Mesopostnotum diamond-shaped, about 1.2 times longer than its width. Anterior margin rounded, lateral and posterior angles arcuate. Metascutum in the mid part strongly narrowed, about 22 times as wide as long in the middle line. Middle indentation in the form of a narrow, deep arc, anterior margin rounded. The lateral part in the form of narrow and blunt convexities directed anteriad. Posterior margin slightly rounded in the lateral part and almost straight in the middle. Metascutellum in the middle part lenticular shape, lateral parts invisible. Parapteron relatively large and oval (Fig. 2A6, A9). Tegula relatively large, with an irregular, slightly triangular shape (Fig. 2A6, A9).

Fig. 2. Whitefly Gregorites bergelmiri Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38972 / ZMUC 902145), Eocene, Danish amber. A1, A10, fore wing; A2, hind wing; A3, A8, antenna; A4, metaleg; A5, genitalia; A6, A9, thorax; A7, abdomen, genitalia, and legs; body in dorsal (A11) and ventral (A12) views. Explanatory drawings (A1–A6), photographs (A7–A12).
Fore wings widening apicad, widest in about 3/5 of the length, about 2.3 times as long as wide (Fig. 2A1, A10, A11, A12). Costal vein arched, anteroapical angle and posteroapical angle arcuate, apical margin arched. Posterior margin rounded basally, further almost straight with delicate depression in the middle part. All margins of the fore wing are covered with small asetose tubercles. Common stem Sc+R+CuA short and thick. Branch Sc+R arcuate, directed towards the apical margin, forked slightly before half the length of the wing. Branch R1 slightly arched basally, further straight, not reaching the margin. Vein Rs straight, about 2 times longer than branch R1, not reaching the margin. Vein CuA arcuate, not reach the margin of the wing. Claval vein CuP straight, ending before half the length of the wing, not reaching the margin. Hind wing wide basally, widening apicad, widest in about 2/3 of the length, about 1.8 times as long as wide (Fig. 2A2, A11, A12). Costal vein arcuate, anteroapical angle and posteroapical angle rounded, apical and posterior margins rounded. All margins of the wing are covered with small asetose tubercles. Branch Sc+R almost straight, directed towards the apical margin, forked before half the length of the wing. Branch R1 arched, not reaching the margin. Branch Rs almost straight, about 1.8 times longer than branch Rs, not reaching the margin.
Profemora not visible (Fig. 2A4, A7, A12). Coxa elongated and slightly conical, trochanters well visible, slightly longer than their width. Metatibia 1.5 times longer than metafemur, tarsus two-segmented, basitmetatarsomere approximately 1.4 times longer than apical metatarsomere, claws long and thin (Fig. 2A4, A12), paronychium not visible. Femora and tibiae covered with fine and fairly, unevenly distributed seta (Fig. 2A4, A7, A12), the longest seta on the apical part of tibia, on the tarsus very short and dispersed seta. There are several long and evenly spaced seta on the inside of the mesotibia, similar to the metatibial comb (Fig. 2A7).
Abdomen relatively slender, longer than the total length of the head and thorax with well visible pregenital segments of similar length (Fig. 2A7, A11, A12). On the ventral side wax fields are visible (Fig. 2A7, A12). Borders between single wax fields are not marked, not visible operculum and lingula. Pygofer about 1.2 times as wide as long in midline, almost quadrangular shape, with a rounded anterior margin (Fig. 2A5, A7). Aedeagus bulbous at the base, distal part invisible, covered with claspers (Fig. 2A5, A7). Claspers elongated and relatively narrow, narrowing posteriad, 1.3 times longer than pygofer (Fig. 2A5, A7, A11, A12). The outer and inner margins of claspers wavy, arched apically, claspers cross at the apical part and curved dorsally.
Stratigraphic and geographic range.—Type locality and horizon only.
Gregorites skadii Drohojowska & Szwedo sp. nov.
Fig. 3.
ZooBank LSID: urn:lsid:zoobank.org:act:24729F11-C24A-43DD-8C C0-693AC9DF480E.
Etymology: After Skadi, the giantess from Norse mythology, representing wilderness of the mountains and hunting.
Holotype: NHMD 38967 [alternative ZMUC 902140], collected by J. Flauensgaard, 21.1.1961, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in dark brown and small sized piece of amber (8×8×5 mm) of cube shape.
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago. Pronotum in lateral part longer than in midline, anterior angles of lateral part strongly rounded, posterior angles acute. Mesopraescutum heart shaped, with slightly incision in anterior margin, clearly separated, slightly wider that its length in the middle line, lateral angles rounded. Mesoscutum relatively wide in midline. Anterior and posterior margins of the middle parts of mesoscutellum mildly rounded, lateral parts almost straight. Anterior margin of mesopostnotum almost straight, lateral angles rounded. Anterior margin of metascutum arched, posterior margin almost straight. Metascutellum in the middle part oval.
Fore wing with vein CuA, branch R1 reaching the margin, branch Rs more than 2 times longer than R1. Hind wing with branch Rs in the form of extension of the Sc+R branch, Rs several times longer than R1 branch.
Description.—Measurements: Table 1. Head from the dorsal side visible only in small part, just above the pronotum, including some ommatidia of the compound eye (Fig. 3A5, A7), from the ventral side head covered with mold (Fig. 3A6, A8). Antennae very poor visible because condition of amber near the antennae not allow to determine the length of individual antennomeres. Pedicel clearly wider than other antennomeres (Fig. 3A6, A9).
Thorax well visible (Fig. 3A4, A5, A7). Pronotum slightly wider than meso- and metathorax, much longer in the lateral parts. Anterior margin widely arcuate, slightly acute in middle, posterior margin acutely incised, lateral parts widened laterad, with rounded anterior angles and acute posterior angles. Mesopraescutum heart shaped, with shallow incision in anterior margin, clearly separated, slightly wider than its length in the middle line, lateral angles rounded, posterolateral margins converging at an angle of 90°. Mesoscutum M-shaped, narrower in the middle, with rounded anterior angles. Mesoscutellum about 5 times wider than its length in the midline. Lateral sections convex, with narrow expansions laterad, anterolateral margin sigmoid, concave, posterolateral margin concave, posterior margin slightly concave in the middle section. Mesoscutellum narrow, band-like, widened and rounded in the middle portion. Anterior and posterior margins of middle portion slightly arched, lateral parts narrow and straight. Mesopostnotum triangular, slightly wider than its length in the midline. Anterior margin is almost straight, lateral angles rounded, the posterolateral margins are almost straight, the posterior angle arcuate. Metascutum strongly narrowed in the middle part, several times wider than its length in the middle line. Anterior margin arcuate, posterolateral margins slightly rounded, lateral parts of metascutum narrow and blunt protuberances directed anteriad. Lateral parts of metascutellum narrow, oval and strongly wider in the middle part, anterior and posterior margins slightly rounded. Lateral parts of metascutellum directed anteriad. Parapteron relatively large and oval (Fig. 3A4, A7). Tegula irregular, subtriangular (Fig. 3A4, A7).
Fore wing widening apicad, the widest in about 3/4 of its length, about 2.5 times as long as wide (Fig. 3A1, A5, A6, A10, A13). Costal vein arched, anteroapical angle arched, apical margin of vein curved, posteroapical angle arcuate. Posterior margin rounded at the base, further almost straight. Margins of fore wing with fine setae. Branch Sc+R straight, directed towards the anteroapical angle, forked at about the middle of the wing length. Branch R1 weakly visible, arcuate, reaching costal vein (Fig. 3A1, A5, A10). Vein Rs straight, 2.1 times longer than vein R1, not reaching margin of wing. Vein CuA straight, weakly marked, slightly curved in the middle part, not reaching the margin of the wing. Claval vein CuP straight, relatively thick and well-marked, not reaching the margin of the wing. Hind wing relatively narrow at the base, widening apicad, the widest in about 3/4 of the length, about 2.3 times as long as wide (Fig. 3A2, A5, A6, A11, A12). The base of the costal vein invisible, covered by the fore wing further straight. Anteroapical angle arcuate, apical margin arched, posterior margin arcuate. The posteroapical angle of hind wing slightly rolled up. Margins covered with fine setae. Branch Sc+R gently arched, directed apicad, forked in the middle of its length. Branch R1 weakly marked, relatively short and straight, not reaching the margin (Fig. 3A2, A5, A11, A12). Branch Rs about 3.3 times longer than branch R1, occurs as an extension of Sc+R branch and not reaching the margin of the wing.
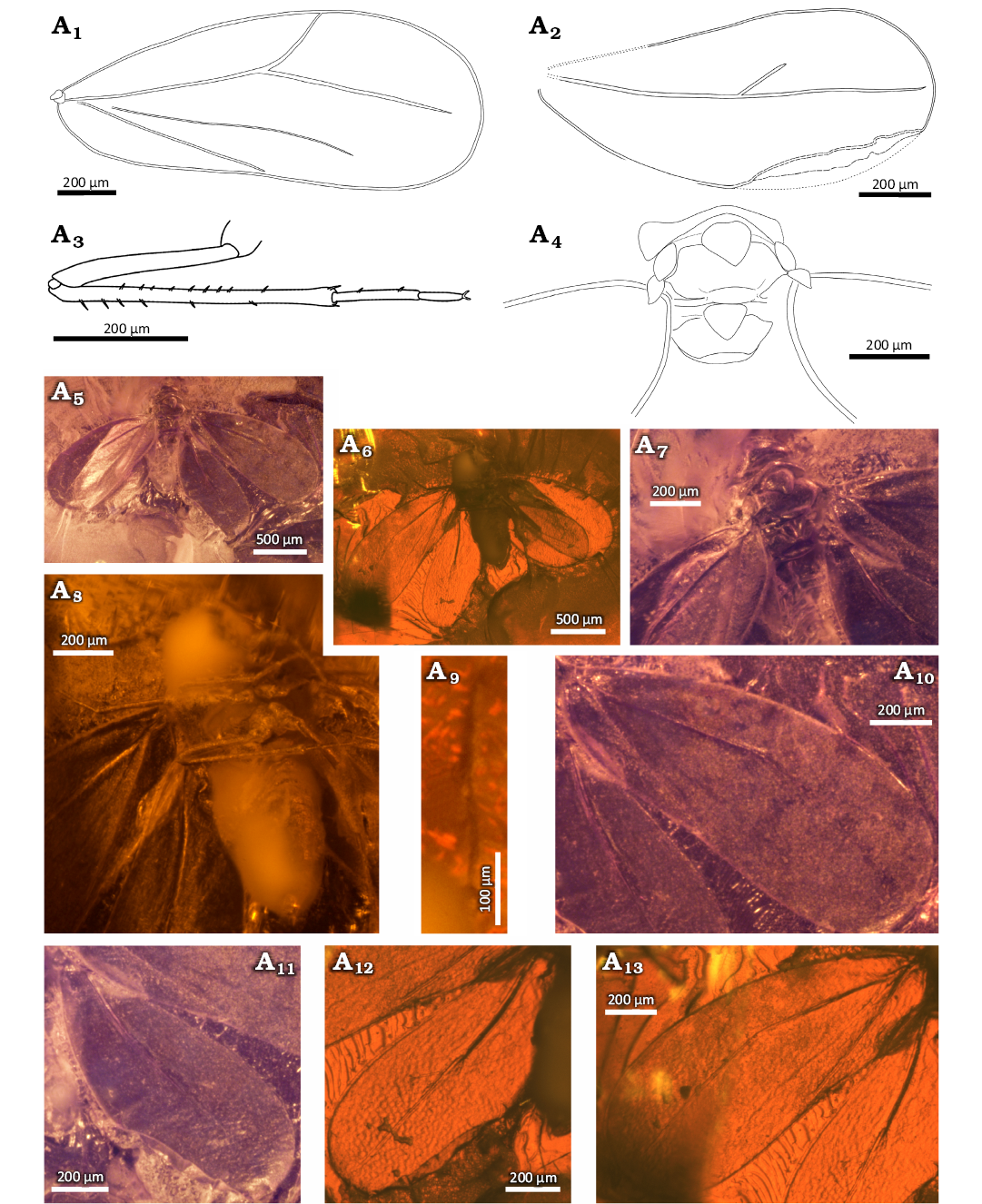
Fig. 3. Whitefly Gregorites skadii Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38967 / ZMUC 902140), Eocene, Danish amber. A1, fore wing; A2, hind wing; A3, metaleg; A4, A7, thorax; body in dorsal (A5) and ventral (A6) views; A8, legs and abdomen; A9, antenna; A10, fore wing dorsal side; A11, hind wing dorsal side; A12, hind wing ventral side; A13, fore wing ventral side. Explanatory drawings (A1–A4), photographs (A5–A13).
Proleg and mesoleg of similar length, metaleg the longest (Fig. 3A3, A6, A8). Coxae elongated and slightly conical, trochanters pronounced, slightly longer than their width. Profemora invisible. Measurements of mesotarsus made at an angle due to the specific positioning of the mesolegs. Metatibia about 1.6 times as long as wide. Tarsi two-segmented. Basimetatarsomere about 1.7 times longer than apical metatarsomere. Claws relatively short and straight, paronychium invisible. Metatibiae covered with relatively long and fairly evenly distributed setae, singular setae visible also on the tarsus.
Abdomen on the dorsal side barely visible (Fig. 3A5), on the ventral side covered with a thin layer of milky veil (Fig. 3A6, A8). Abdomen oval and longer than the total length of the head and thorax. Gender cannot be determined, probably female due to the characteristic triangular shape the apical part of the abdomen (visible from the lateral side) and lack of claspers and aedeagus (Fig. 3A6, A8).
Stratigraphic and geographic range.—Type locality and horizon only.
Gregorites bestlae Drohojowska & Szwedo sp. nov.
Fig. 4.
ZooBank LSID: urn:lsid:zoobank.org:act:F6404564-8183-4529-A7BD- 5E27B8EEAD32.
Etymology: After Bestla, the giantess from Norse mythology, the name means “thorn of evil”.
Holotype: NHMD 38962 [alternative ZMUC 902135], collected by C.V. Henningsen, 16.5.1957, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in clear dark brown and very small sized cube shaped piece of amber (6×3×4 mm).
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago. Antennomeres 4th and 6th equal in size. Anterior margin of mesopostnotum with delicate depression in the middle part, anterior angles rounded, posterior angle arcuate. Wide arched incision in the middle part of metascutum, posterolateral margins strongly rounded. Fore wing with vein CuA present, branch Rs more than 2 times longer than branch R1, hind wing with branch Sc+R not reaching margin.
Description.—Measurements: Table 1. Head invisible from the ventral and dorsal side due to the specific arrangement of the specimen and numerous impurities in amber (Fig. 4A8), part of head visible in the lateral view (Fig. 4A7, A10, A13, A14). Compound eyes not bulging, not divided, ommatidia in equal size (Fig. 4A10, A13). Relatively large lateral ocelli located above the compound eyes (Fig. 4A10, A13).
Antennae seven-segmented (Fig. 4A3, A13). Scapus not visible, pedicel visible partially, longer than its width, wider at the apical part. Third antennomere the longest, about 3 times longer than 4th one. Antennomeres 4th and 6th equal in size, 5th one 1.2 times longer than 4th and 6th antennomeres; 7th antennomere about 1.2 times shorter than 6th, tapering towards apex.
From the dorsal side only a part of thorax is visible (Fig. 4A6, A8, A9). Mesopostnotum triangular, almost as wide as long in midline. Anterior margin with delicate depression in the middle part, anterior angles rounded, posterolateral margin long, almost straight, posterior angle arcuate. Metascutum partly covered by wings, in the middle part visibly narrowed, at least several times as wide as long in midline. Anterior margin with distinctly depressions (probably taphonomic artifact), incision in the middle part wide arched, lateral part in the form of narrow and blunt protuberances gently directed anteriad, posterolateral margins gently rounded. Metascutellum narrow laterally, much wider in the middle part with anterior and posterior margins rounded.
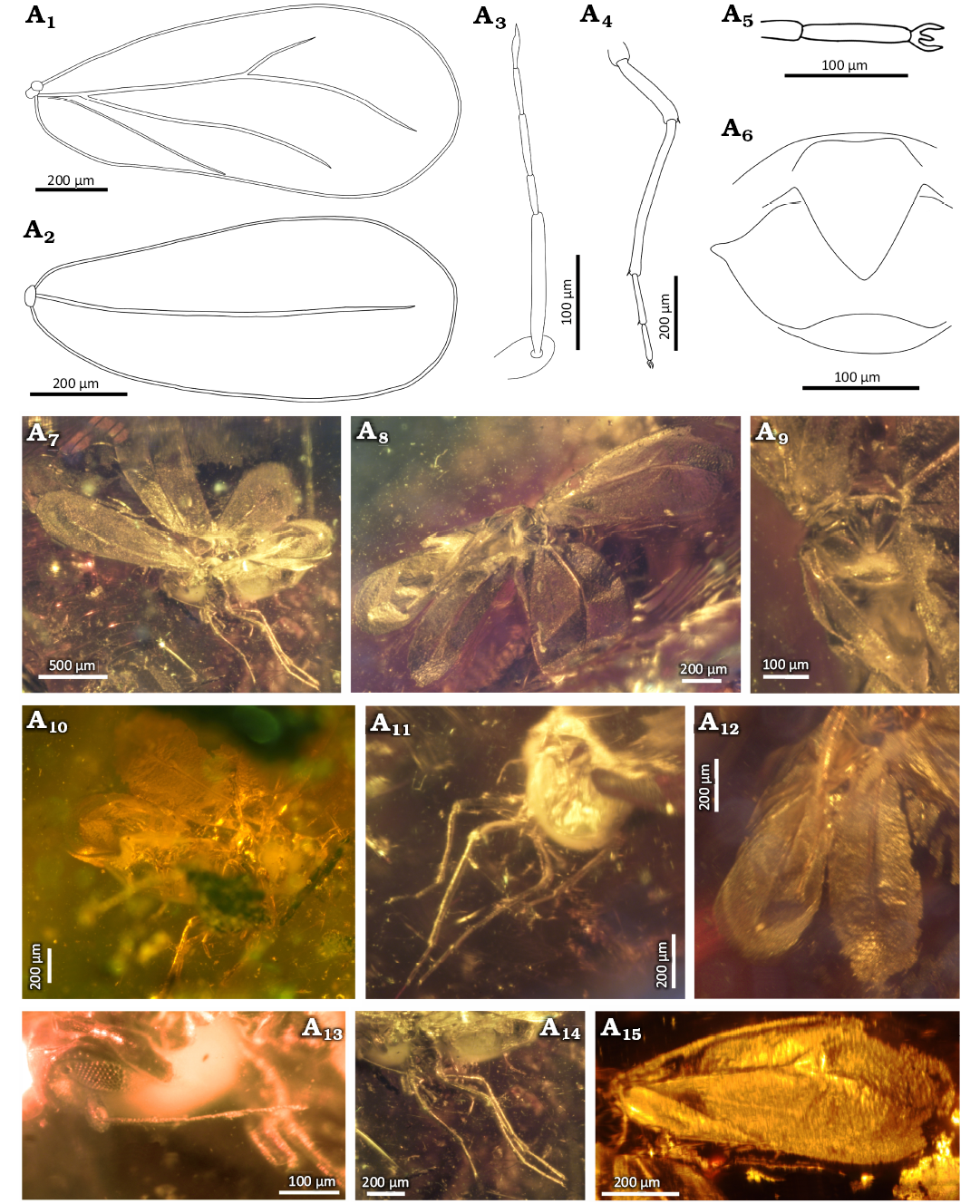
Fig. 4. Whitefly Gregorites bestlae Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38962 / ZMUC 902135), Eocene, Danish amber. A1 A15, fore wing; A2 A12, hind wing; A3, antenna; A4, metaleg; A5, mesotarsus; A6, A9, thorax; body in lateral (A7), dorsal (A8), and lateroventral (A10) views; A11, legs; A13, head and antenna; A14, legs, wax plates. Explanatory drawings (A1–A6), photographs (A7–A15).
The fore wing widening apicad, widest in about 3/4 of the length of the wing, about 2.3 times as long as wide (Fig. 4A1, A7, A8, A15). Costal vein almost straight at base, further slightly arched, anteroapical angle and posteroapical angle arcuate, apical margin arched. Posteroapical margin rounded basally, further almost straight. Common stem Sc+R+CuA short and thick. Branch Sc+R straight, directed towards the anteroapical angle, forked in the middle of the length of the wing. Branch R1 arcuate, not reaching a margin of wing. Branch Rs arcuate, 2.3 times longer than branch R1, not reaching to margin of wing. Vein CuA wavy, extending at an acute angle from the common stem Sc+R+CuA, not reaching the margin of the wing. Claval vein CuP straight, reaching almost half the length of the wing, not reaching the margin. Hind wing slightly distorted at the base, gradually widening apicad, widest in about 3/4 of length, about 2.6 times as long as wide (Fig. 4A2, A7, A8, A10, A12). Costal vein arcuate at the base, further almost straight. Anteroapical angle arcuate, apical margin arched, posteroapical angle rounded, posterior margin arched. Branch Sc+R slightly arcuate, not reaching the margin of wing.
Proleg invisible except for the apical tarsomere (Fig. 4A4, A7, A11, A14). Mesocoxa and metacoxa invisible, meso- and metatrochanters poorly marked. Meso- and metafemora the same length, metatibia at least 1.4 times longer than femur, tarsus two-segmented. Basimetatarsomere about 1.4 times longer than apical metatarsomere, claws distinct, relatively long and narrow (Fig. 4A4, A5, A14). Mesoparonychium visible reaching about half the length of the claw, similar in width to it (Fig. 4A5, A14). Setae short and few, the longest on apical part of legs (Fig. 4A4, A7, A11, A14). On the inner side of metaleg hardly visible tibial combs composed of several long and relatively even distributed seta (Fig. 4A11).
Abdomen invisible from the dorsal side, covered with wings, the ventral side of abdomen invisible (Fig. 4A8). From the lateral side abdomen seems massive, longer than the total length of the head and thorax (Fig. 4A7, A10, A14). From the lateral side only two wax plates well visible, the other two damaged (Fig. 4A7, A14). The wax plates large, rectangular with rounded angles and slightly wider than their length. Gender cannot be determined, probably female due to the characteristic triangular shape the apical part of the abdomen (visible from the lateral side) and lack of claspers and aedeagus (Fig. 4A10, A11, A14).
Stratigraphic and geographic range.—Type locality and horizon only.
Gregorites thrymi Drohojowska & Szwedo sp. nov.
Fig. 5.
ZooBank LSID: urn:lsid:zoobank.org:act:0B065409-1718-40BF-AB15- F8980DF3BD3E.
Etymology: After Thrym, the giant in Norse mythology, king of the jǫtnar (giants) ruling Jötunheimr.
Holotype: Male, NHMD 38836 [alternative ZMUC 902009], collected by C.V. Henningsen, 1.2.1969, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in dark brown and extremally small sized piece of amber (3×2×2 mm) of cubical shape. Previously labelled as Psyllidae.
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago, male. Fourth antennomere almost the same length as 5th, antennomeres 6th and 7th the same length, antennomere 7th without narrowing in distal part. Branches R1, Rs, and CuA reaching margin of fore wing, claval vein CuP longer than half length of wing, branch Rs in fore wing and hind wing almost 2 times longer than branch R1. Hind wing with atypical shape, strongly narrowed basally, strongly widened apically. Claspers more than 2 times longer than pygofer, the inner side of claspers with a convexity in the middle, not crossing apicad. Almost all part of body covered by very tiny, densely dispersed hairs.
Description.—Measurements: Table 1. The head from the ventral side covered with mold and gas bubbles (Fig. 5A8, A9, A10, A11), from the dorsal side completely invisible (Fig. 5A7, A13). From the ventral side a part of short, massive rostrum is visible (Fig. 5A8, A10, A11) reaching to the mesocoxa. Rostrum covered with fine hairs. Compound eyes undivided (Fig. 5A9). Relatively big lateral ocelli are situated above compound eyes, visible from the lateral side (Fig. 5A9). Antennae seven-segmented (Fig. 5A3, A11). First antennomere (scapus) is invisible. Second antennomere (pedicel) visible only in the upper part, about 3.5 times wider than the width of 3rd one. Third antennomere the longest, about 2.5 times longer than 4th. Fourth antennomere almost the same length as 5th. Antennomeres 6th and 7th the same length, shorter than antennomeres 4th and 5th. Antennomere 7th is bluntly terminated, without narrowing in distal part. All antennomeres covered with very tiny hairs (Fig. 5A11).
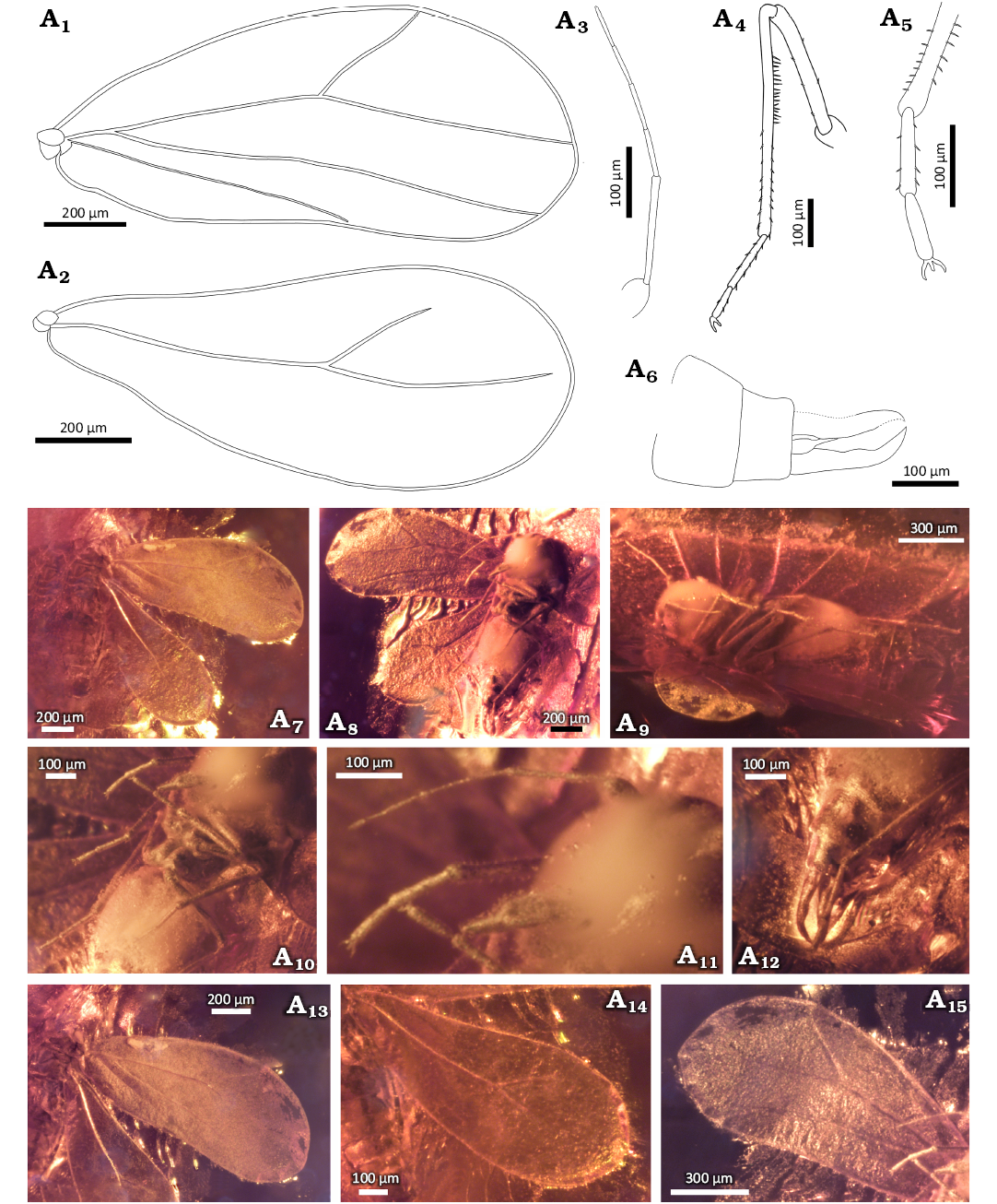
Fig. 5. Whitefly Gregorites thrymi Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38836 / ZMUC 902009), Eocene, Danish amber. A1, fore wing; A2, A14, hind wing; A3, antenna; A4, metaleg; A5, protarsus; A6, genitalia; body in dorsal (A7), ventral (A8), and lateral (A9) views; A10, legs; A11, antenna, protarsus, and rostrum; A12, genitalia with pregenital segments; fore wing dorsal side (A13) and ventral side (A15). Explanatory drawings (A1–A6), photographs (A7–A15).
Thorax not visible (Fig. 5A7, A13).
Fore wing widening apicad, the widest in about 2/3 of the length, about 2.2 times longer than its width (Fig. 5A1, A7, A8, A9, A13, A15). Costal vein strongly curved. Anteroapical angle widely rounded, apical margin of the wing is arcuately curved, the posteroapical angle is arched. Posterior margin of the wing is arcuate at the base, further almost straight, the middle part with a slight depression. Margins of the wing are covered with fine setae (Fig. 5A13, A15). Common stem Sc+R+CuA short and thick. Stem S+R straight, forked apicad of half of fore wing length, branch R1 slightly arched, reaching to the fore wing margin (Fig. 5A1, A7, A9, A13, A15). Branch Rs straight, about 1.9 times longer than branch R1, probably reaching apical margin of the wing (Fig. 5A1, A7, A15). Vein CuA straight, extending at an acute angle from the common stem Sc+R+CuA, almost parallel to the Rs branch, probably reaching to the fore wing margin (Fig. 5A1, A15). Claval vein CuP thin and straight, not reaching to the margin (Fig. 5A1, A8, A13). Hind wing narrow at the base, clearly widening apicad, widest in about 3/5 hind wing length, about 2.3 times longer than wide (Fig. 5A2, A7, A8, A14). Costal vein slightly arched at the base, then almost straight. Anteroapical angle broadly rounded, apical margin arcuate, posteroapical angle broadly rounded, posterior margin arcuate. Margins of hind wing covered with fine setae (Fig. 5A14). Sc+R branch slightly arched at the base, further straight, forked at half of hind wing. Branch R1 gently arched, not reaching to the margin. Branch Rs arched, about twice as long as branch R1, directed towards the anteroapical angle, not reaching to the margin.
Proleg the shortest and metaleg the longest (Fig. 5A4, A8, A9, A10, A11). Coxae elongated and conical. Trochanters slightly wider than their length. Femora shorter than tibiae. Tarsi two-segmented, basitarsomere significantly longer than apical tarsomere. The claws relatively long and thin (Fig. 5A4, A5, A10, A11). Paronychium of proleg and mesoleg 2 times shorter than the claw, similar in width to the claw (Fig. 5A5, A10, A11). Paronychium of metaleg invisible. Metatibia 1.7 times longer than mesofemur. Basimetatarsomere about 1.4 times longer than the apical metatarsomere. On ventral side of metatibia well visible metatibial comb consisting of dozen or so, dispersed, long setae (Fig. 5A4, A10). Femora, tibiae and tarsi of pro-, meso-, and metalegs densely covered with fine, but distinct rows of tiny setae (Fig. 5A4, A5, A10, A11). Abdomen on ventral side covered with gas bubble and mold (Fig. 5A8, A9, A10), only last pregenital segments and male genitalia visible (Fig. 5A6, A8, A12). Pygofer about 1.6 times as high as long in midline (Fig. 5A6, A12). Aedeagus with bulbous base, thin and straight in the middle, apical portion invisible (obscured by claspers) (Fig. 5A6, A12). Claspers elongate, gradually narrowing apicad, twice as long as pygofer (Fig. 5A6, A8, A12). The outer part of claspers is slightly arched, the inner with a convexity in the middle. Claspers do not crossed at apex, tapered in apical half, with apices pointed and curved dorsally.
Almost all part of body covered by very tiny, densely dispersed hairs, giving the impression of “mossiness” (Fig. 5A8, A10, A11).
Stratigraphic and geographic range.—Type locality and horizon only.
Gregorites halogii Drohojowska & Szwedo sp. nov.
Fig. 6.
ZooBank LSID: urn:lsid:zoobank.org:act:BD32F78A-AD06-4C4D-A1 CC-C8C536B50958.
Etymology: Specific name after Hálogi, a jötunn (giant) and the personification of fire in Norse mythology.
Holotype: Male, NHMD 38960 [alternative ZMUC 902133], collected by C.V. Henningsen, 16.5.1956, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in dark brown and small sized piece of amber (12×6×2 mm) of flat cube shape.
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago, male. Pedicel relatively short and thick with heart-shaped apical part and two short setae on lateral margin 4th antennomere longer than 5th, 6th antennomere shorter than 7th. Fore wing with vein CuA present. Apical part of claspers strongly curved dorsad, lateral margin with shallow indentation at base, medial margin with convexity in the middle part, claspers not crossed at apex. Aedeagus funnel-shaped at the base, further straight, with apex exceeding 3/4 of length of claspers.
Description.—Measurements: Table 1. Head from the dorsal side invisible (Fig. 6A7). The median incision in the anterior margin of head clearly visible from the ventral side (Fig. 6A6, A8, A9, A14). Compound eyes relatively large and bulging, not divided, ommatidia equal in size (Fig. 6A9, A14). Lateral ocelli invisible. Rostrum short and massive, reaching to the mesocoxa (Fig. 6A6, A8, A9, A14).
Antennae seven-segmented (Fig. 6A3, A9, A12, A14). Scapus about 1.6 times as wide as long, lateral margins gently rounded. Pedicel relatively short (compared to the 2nd antennomere of other described species) and massive, wider at the apical part, about 1.6 times as long as wide with anterior part heart-shaped and two short seta on lateral margin. Third antennomere the longest, about 1.5 times longer than 4th. Fourth antennomere about 1.2 times longer than 5th, 6th antennomere the shortest, about 2.2 times shorter than 5th, 7th antennomere 1.5 times longer than 6th, and tapering towards apex.
Thorax on the dorsal side completely covered by wings and an opaque layer of amber (Fig. 6A7). Fore wings visible only from the dorsal side. Fore wing gradually widening towards the apex, widest at about 3/4 of the length, almost 2 times as long as wide (Fig. 6A1 A7, A13). Costal vein arcuate curved, anteroapical angle broadly rounded, apical margin and posteroapical angle rounded. Posterior margin of fore wing arched at the base, further almost straight. Common stem Sc+R+CuA short and thick. The Sc+R branch slightly arched, directed towards the anteroapical angle of the wing, forked before half its length. Branches R1 and Rs slightly arched, both not reaching a margin, Rs about 1.7 times as long as R1.
Vein CuA slightly wavy, protruding at an acute angle from the common stem Sc+R+CuA, not reaching the margin of wing. Claval vein CuP thin, slightly arched, reaching less than half the length of the wing, not reaching the margin. Hind wings invisible, covered by the fore wings (Fig. 6A7).

Fig. 6. Whitefly Gregorites halogii Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38960 / ZMUC 902133), Eocene, Danish amber. A1, A13, fore wing; A2, A12, antenna; A3, metaleg; A4, mesotarsus; A5, genitalia; A6, A8, body in lateroventral view; A7, body in dorsal view with fore wings; A9, A14, head; A10, genitalia, pregenital segments; A11, abdomen. Explanatory drawings (A1–A5), photographs (A6–A14).
Prolegs the shortest, metalegs the longest, 1.2 times longer than mesolegs (Fig. 6A3, A4, A6, A8). Procoxae slightly conical, trochanters poorly marked, almost twice as wide as long, profemora shorter than tibia, protarsus two-segmented, basiprotarsomere visible, longer than apical protarsomere. Metatibia 2 times longer than metafemur. Basimetatarsomere about 1.6 times longer than apical metatarsomere. Total length of metatarsus similar to length of metafemur. Claws long, narrow and hooked, apical upwards (Fig. 6A3, A4, A6, A8). Paronychium invisible. Tibiae and tarsi covered with rows of setae, femora smooth. The comb of setae visible on the inner side of metatibia, consisting of long and evenly distributed setae (Fig. 6A6).
The abdomen invisible from the dorsal side, covered by wings and not transparent, cracked layer of amber (Fig. 6A7). From the ventral side abdomen seems quite massive, slightly longer than the total length of the head and thorax (Fig. 6A6, A8, A11). Three wax plates visible (Fig. 6A11). First wax plate partially visible, the same width as the 2nd. Second and 3rd wax plate as long as wide, with rounded edges. Pygofer concave in basal 2/3 of the length (Fig. 6A5, A10, A11). Aedeagus long (compared to the length of claspers), funnel-shaped at the base, further thin and straight, length of aedeagus reaches up to about 3/4 of the length of claspers (Fig. 6A5, A10, A11). Claspers elongated, broad, narrowed in the distal part, not crossed at apex, curved dorsad (Fig. 6A5, A10, A11). Lateral margin with slightly indentation at base, further arcuated. Medial margin with convexity in the middle part. Apical part of claspers covered with single setae.
Stratigraphic and geographic range.—Type locality and horizon only.
Gregorites ymiri Drohojowska & Szwedo sp. nov.
Fig. 7.
ZooBank LSID: urn:lsid:zoobank.org:act:F8035251-485E-4750-9D43- 509D4CDAFC10.
Etymology: After Ymir—in Norse mythology the first giant emerged from Ginnungagap.
Holotype: NHMD 38974 [alternative ZMUC 902147], collected by A.K. Andersen, 28.3.1968, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in clear light brownish and moderate sized piece of amber (20×10 mm) of irregular shape.
Type locality: Danish amber; western coast of Denmark.
Type horizon: Lutetian–Priabonian, Eocene.
Diagnosis.—Imago. Posterior margin of pronotum slightly arched, anterolateral angles acute, posterolateral angles rounded. Mesopraescutum wider than its length in the midline, anterior margin slightly curved, lateral angles rounded, the posterolateral margin gently curved. Anterior angles of mesoscutum strongly rounded, anterolateral margins with clear recesses, posterolateral margins slightly convex and curved, posterior margin with rounded recess. Anterior and posterior margins of the middle part of mesoscutellum mildly rounded, lateral parts almost straight. Anterior margin of mesopostnotum straight, lateral angles rounded, posterior angle curved. Metascutum with median indentation deep and curved, anterior margins rounded, posterolateral margins much longer than the anterior ones, rounded, side parts facing laterally, posterior margin with rounded recess. Anterior margin of metascutellum strongly rounded. Fore wing with vein CuA, branch Rs 2 times longer than branch R1.
Description.—Measurements: Table 1. Head partially visible (Fig. 7A7, A9). Compound eyes relatively large, anteriorly incised (Fig. 7A9). Ommatidia in the dorsal part smaller and densely arranged, in the ventral part are larger and sparsely arranged. Lateral ocelli not visible. Coronal suture not visible. The amber near the specimen is muddy, not transparent and not allowing to count the number of antennomeres (Fig. 7A4, A6, A9). Scapus is relatively large (Fig. 7A9), pedicel large and massive, widening towards apex. Third antennomere 2 times longer than 4th. Other antennomeres not visible.
Thorax visible (Fig. 7A3, A7). Pronotum wider than mesothorax and metathorax, seems to be also wider than the head. Lateral parts longer than length of pronotum in midline. Anterior margin slightly with acute protrusion in the middle, posterior margin slightly arcuate. Anterolateral angles acute, posterolateral angulately rounded. Triangular mesopraescutum clearly separated, wider than long in the midline. Anterior margin of mesopraescutum slightly arched, lateral angles rounded, posterolateral margins slightly arched, converging at an angle of 90°. M-shaped mesoscutum, narrower in the middle, with rounded anterior angles, about 6.8 times as wide as long in midline. Lateral parts form relatively long, narrow and blunt expansions. Anterolateral margins with clear indentations, posterolateral margins slightly convex and arched. Posterior margin concave in the middle. Mesoscutellum forms a narrow band, clearly wider and rounded in the middle part, the anterior and posterior margins of the mid part slightly rounded, lateral parts narrow and almost straight. Triangular mesopostnotum, slightly wider than its length in the midline, anterior margin straight, lateral angles rounded, posterolateral margins arcuate, posterior angle arched, triangular and prolonged caudad depression the midline visible. Metascutum partly covered by wings, strongly narrowed in the middle, several times as wide as long in the midline. Middle deep and arcuate indentation, anterior margins rounded, the lateral parts form narrow and blunt protuberances directed laterad, posterolateral margins longer than the anterior ones, rounded, posterior margin with a rounded indentation. Metascutellum narrow laterally, clearly wider in the middle, anterior margin rounded, posterior arcuate. Lateral bands narrow directed anteriad. The mesopostnotum, mesoscutellum and metascutellum measurements included only the middle part, without narrow lateral bands. Parapteron large and oval (Fig. 7A3, A7). Tegula relatively large, of irregular shape (Fig. 7A3, A7).

Fig. 7. Whitefly Gregorites ymiri Drohojowska & Szwedo gen. et sp. nov., holotype (NHMD 38974 / ZMUC 902147), Eocene, Danish amber. A1, fore wing; A2, hind wing; A3, A7, thorax; A4, A6, antenna; body in dorsal (A5) and anterior (A9) views; A8, wings. Explanatory drawings (A1–A3), photographs (A4–A9).
Fore wing widening apicad, the widest in about 2/3 of the length, about 2.3 times as long as wide (Fig. 7A1, A5, A8). Costal vein arcuate, anteroapical and posteroapical angles arcuate, apical margin arched. Posterior margin rounded basally, further straight in the middle part with slightly depression. Common stem Sc+R+CuA weakly visible. Branch Sc+R straight, directed apicad, forked about half the length of the wing. Branch R1 straight, not reaching the margin. Branch Rs almost straight, about 2 times longer than the branch R1, not reaching the margin. Vein CuA curved in the middle part, departing at an acute angle from the common stem Sc+R+CuA, not reaching the margin, almost parallel to the branch Rs. Claval vein CuP straight, not reaching the margin. Hind wing partially covered by the fore wings, however their shape can be described (Fig. 7A2, A5, A8). Hind wing widening apicad, the widest in about 3/5 of the length, about 2.6 time as long as wide. Basal part of the costal vein invisible, the anteroapical and posteroapical angles arcuate, the apical and posterior margins arched.
Legs visible only partly (Fig. 7A5, A9). Abdomen invisible. Gender cannot be determined (Fig. 7A5).
Remarks.—The specimen is placed right at the edge of amber, which had a significant impact on the poor quality of observations and photos taken.
Stratigraphic and geographic range.—Type locality and horizon only.
Aleyrodidae gen. et sp. indet. 1
Fig. 8.
Material.—Male, NHMD 38970 [alternative ZMUC 902143], Danish amber; western coast of Denmark; Lutetian–Priabonian, Eocene, collected by C.V. Henningsen, 19.6.1964, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in clear brownish and small piece of amber (7×5×2 mm) of flat cube shape. Specimen located right on the surface of amber. The condition of some fragments of the dorsal side of the body suggests that they protruded beyond the piece of amber and decomposed.
Description.—Measurements: Table 1. Head from the dorsal side destroyed (Fig. 8A7, A8, A12). Compound eyes large and bulging, not divided with big and equal in size ommatidia (Fig. 8A9, A10). Lateral ocelli not visible. Antennae seven-segmented (Fig. 8A3, A7, A12). Scapus visible partially, relatively short with angular margins. Pedicel large and massive, wider at the apical part, about 2.4 times as long as wide. Third antennomere the longest, about 4 times longer than 4th. Fourth antennomere the same length as 6th, antennomeres 5th and 7th equal in size and slightly longer than antennomeres 4th and 6th, 7th antennomere tapering apicad.
Thorax in dorsal view not preserved (Fig. 8A7, A8). Only a metascutellum is visible, with a lenticular median part and narrow lateral parts.
Fore wing only as an imprint on the amber surface (Fig. 8A1, A8, A9). The visible shape of the wing and a part of the branch Rs remained. The impressed fore wing widening apicad, the widest in about 3/4 of the length, about 2.2 times as long as wide. Costal vein imprint invisible at the base, further straight. Anteroapical angle arcuate, apical margin arched, posteroapical angle arcuate, posterior margin invisible at the base, further arcuate. Hind wing narrow at the base, widening apicad, the widest in about ⅘ of the length, about 2.4 times as long as wide (Fig. 8A2, A7, A8, A9, A11). Costal vein arcuate at the base, further almost straight, anteroapical angle arcuate, apical margin arched, posteroapical angle rounded, posterior margin arcuate. Branch Sc+R straight, not reaching margin.

Fig. 8. Aleyrodidae gen. et sp. indet. 1 (NHMD 38970 / ZMUC 902143), Eocene, Danish amber. A1, fore wing; A2, A11, hind wing; A3, A4, A12, antenna; A5, genitalia; A6, genitalia, pregenital segments; body in dorsal (A7, A8,) and ventral (A9)views; A10, legs and head; A13, abdomen with legs. Explanatory drawings (A1–A5), photographs (A6–A13).
Metaleg about 1.3 times longer than mesoleg (Fig. 8A4, A9, A10, A13). Metacoxa elongated, trochanters poorly visible. Metatibia about 1.2 times longer than metafemora. Tarsi two-segmented. Basimetatarsomere about 1.3 times longer than apical tarsomere. Claws relatively short and thin (Fig. 8A4, A13). Paronychium poorly visible, about half the length of the claw (Fig. 8A13). Metatibia and metatarsus covered with relatively evenly dispersed, fine seta (Fig. 8A4, A7, A9, A10, A13). Metafemora without seta. The tibial combs consist of 13 long and evenly distributed seta on the inner side of the metatibia (Fig. 8A4, A13).
Abdomen collapsed on the dorsal side (Fig. 8A7, A8), from the ventral side partly covered by a cloudy part of amber (Fig. 8A9, A10, A13). Abdomen relatively slim, strongly narrowed at the thorax, longer than the total length of the head and thorax. Pygofer slightly wider than its length in the midline, almost quadrangular in shape (Fig. 8A5, A13, A6). Aedeagus bottle in shape at the base, then straight, with apical part not visible (hidden behind the claspers) (Fig. 8A5, A13, A6). Claspers elongated and relatively wide, slightly narrowed distally, as long as the pygofer (Fig. 8A5, A13, A6). The outer margin slightly arched, the inner margin with convexity in the middle. Claspers overlapping at the apical part and curved dorsad.
Aleyrodidae gen. et sp. indet. 2
Fig. 9A.
Material.—Female, NHMD 38971 [alternative ZMUC 902144], Danish amber; western coast of Denmark; Lutetian–Priabonian, Eocene, collected by C.V. Henningsen, 1.7.1966, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in clear light yellowish and small sized piece of amber in flat cube shape (10×8×3 mm). Specimen located near edges of amber, which significantly hindered observation and taking pictures.
Description.—Measurements: Table 1. Head invisible (Fig. 9A4, A8). Antennae seven-segmented (Fig. 9A3, A5). Scapus not visible, pedicel large and massive, tapering towards apex, at least 1.7 times as long as wide. Third antennomere the longest, about 4 times longer than 4th. Fourth and 6th antennomeres equal in size, antennomere 5th the same length as 7th, about 1.2 times longer than antennomeres 4th and 6th. Antennomere 7th tapering towards apex.
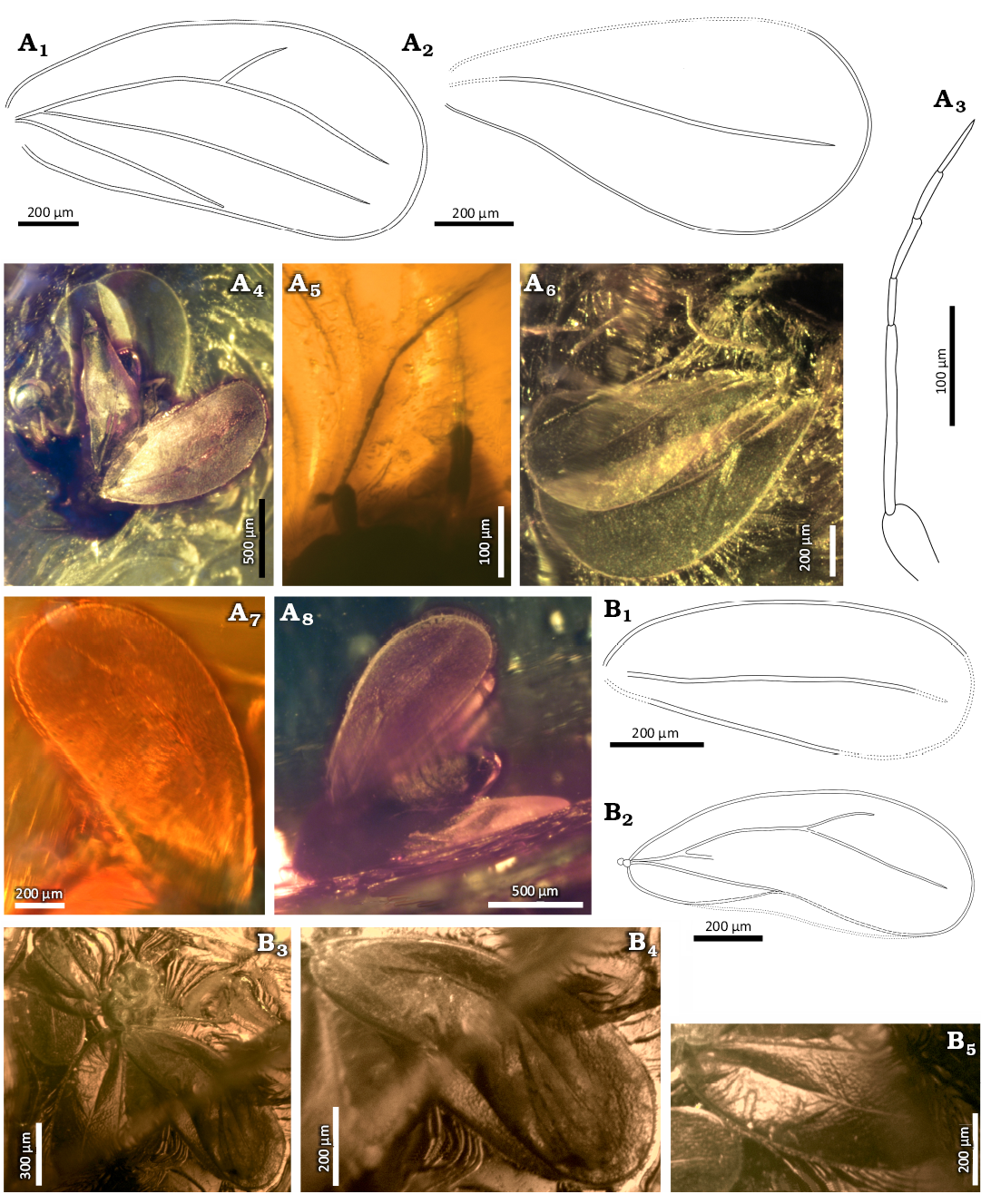
Fig. 9. Aleyrodidae gen. et sp. indet. 2 and 3 (respectively), Eocene, Danish amber. A. NHMD 38971 / ZMUC 902144. A1, fore wing; A2, hind wing; A3, A5, antenna; body in lateral (A4) and posteroventral (A8) views; A6, wings; A7, fore wings. B. NHMD 38973 / ZMUC 902146. B1, B4, fore wing; B2, B5, hind wing; B3, body in laterodorsal view. Explanatory drawings (A1–A3, B1, B2), photographs (A4–A8, B3–B5).
Fore wing widening apicad, the widest in about 3/4 of the length, about 2.1 times as long as wide (Fig. 9A1, A4, A6, A7, A8). Costal vein rounded at the base, further arcuate, anteroapical angle arcuate, apical margin arched, posteroapical angle arcuate. Posterior margin rounded at the base, then straight. Several relatively long setae visible on the apical edge of the fore wing. Common stem Sc+R+CuA short and thick (Fig. 9A1, A4). Branch Sc+R arched, directed towards the anteroapical angle, forked about half the length of the wing. Branch R1 (Fig. 9A1, A6, A8) weakly visible, slightly arched, not reaching the margin. Branch Rs slightly curved in the middle part, about 2.3 times longer than the branch R1, not reaching the margin. Vein CuA almost straight, slightly curved in the middle part, departing at an acute angle from the common stem Sc+R+CuA, almost parallel to the branch Rs, not reaching the margin (Fig. 9A1, A4, A8). Claval vein CuP straight, not reaching the margin (Fig. 9A1, A4). Hind wing widening apicad, the widest in about 2/3 of the wing length, at least 1.9 times as long as wide (Fig. 9A2, A4, A6). Anterior margin invisible, anteroapical angle arcuate, apical margin arched, posteroapical angle rounded, posteriori margin straight with a slight depression in the middle part. Branch Sc+R slightly curved in the middle, not reaching the margin.
Legs visible partly only (Fig. 9A6), thorax not visible (Fig. 9A4, A6, A8).
Abdomen visible fragmentarily, mainly in the distal part (Fig. 9A4, A8). Gender cannot be determined. Probably female due to characteristic triangular shape of apical part of the abdomen (visible from the lateral view) and the lack of claspers or aedeagus (Fig. 9A4, A8).
Aleyrodidae gen. et sp. indet. 3
Fig. 9B.
Material.—Specimen NHMD 38973 [alternative ZMUC 902146], Danish amber; western coast of Denmark; Lutetian–Priabonian, Eocene, collected by C.V. Henningsen, 20.3.1975, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in moderate sized piece of amber (22×18 mm) of irregular shape. The piece of amber has an extremely dark colour, and the part of amber near the inclusions is strongly cracked, which made observation difficult.
Description.—Measurements: Table 1. Head from the lateral side visible (Fig. 9B3). Compound eyes large, not bulging (Fig. 9B3), ommatidia relatively small and equal in size. Lateral ocelli large, located above the compound eyes (Fig. 9B3). Antennae completely invisible.
Left fore wing folded in half, the right fore wing crumpled and a crack in the amber structure passes through the centre (Fig. 9B1, B3, B4). Fore wing widening apicad, the widest in about 3/4 of the length, about 2.5 times as long as wide. Costal vein arched, anteroapical angle and posteroapical angle arcuate, apical margin arched, posterior margin rounded basally, further invisible due to this part of wing crumpled. Common stem Sc+R+CuA short and thick. Stem Sc+R appears arcuate (however, a cut of the wing surface may have contributed to the displacement of the use), directed towards the anteroapical angle and forked in the middle of the length of the wing. Branch R1 arcuate, not reaching the margin. Branch Rs straight, about 2.2 times longer than the branch R1, not reaching margin. Vein CuA visible only in a small, basal part, comes away the common branch Sc+R+CuA at an acute angle. Claval vein CuP straight, ending invisible. Hind wing widening apicad, widest in about 2/3 of the length, about 2.4 times as long as wide (Fig. 9B2, B3, B5). Costal vein arched, anteroapical angle slightly arched, apical margin and posteroapical angle invisible due to a crack in amber passing through the tip of the wing. Posterior margin basally invisible, further straight. Stem Sc+R relatively straight, reaching apical part of the wing, stem end invisible. Legs invisible, except from part of tibia and tarsi one of the indeterminate legs (Fig. 9B3). Thorax invisible (Fig. 9B3). The abdomen is visible fragmentarily, in the form of small part of several segments between the wings. Gender cannot be determined (Fig. 9B3).
Aleyrodidae gen. et sp. indet. 4
Fig. 10.
Material.—Specimen female, NHMD 38963 [alternative ZMUC 902136], Danish amber; western coast of Denmark; Lutetian–Priabonian, Eocene, collected by C.V. Henningsen, 19.6.1964, deposited in Amber Collection in Natural History Museum of Denmark. Imago of whitefly is included in clear brownish and small sized piece of amber (6×5×2 mm) of flat cube shape.
Description.—Measurements: Table 1. The head from the dorsal side invisible (Fig. 10A5), from the ventral side only a small part of the compound eye and the antennae are visible (Fig. 10A4, A8). The lack of proper focus and the presence of mold and distortion did not allow to description of the length of separate antennomeres (Fig. 10A8). Pedicel is relatively large, twice as long as wide, the last antennomere distinctly tapering toward apex.
Thorax from the dorsal side invisible, covered by wings and numerous small cracks in amber (Fig. 10A5). Wings covered with impurities limiting visibility (Fig. 10A4, A5, A7). The base of fore wings invisible (Fig. 10A1, A4, A5, A7). The fore wing widening apicad, at least 1.9 times as long as wide, the widest in about 3/4 of the length. Visible part of the anterior margin arcuate, anteroapical angle rounded, apical margin rounded too, posteroapical angle arched, visible posterior margin arcuated further straight. Common stem Sc+R+CuA thick. Branch Sc+R straight, directed towards the anteroapical angle. Branch R1 and the end of branch Rs invisible. Vein CuA straight with invisible ending. Claval vein CuP thin and straight with invisible ending. Hind wing narrow at the base, gradually widening apicad, 2.8 times as long as wide, the widest in about 3/4 of its length (Fig. 10A2, A4, A5, A6). Costal vein slightly arched at the base, further almost straight, anteroapical angle arcuate, apical margin arched, posteroapical angle arched, posterior margin arched, too. Branch Sc+R long and almost straight, in distal part slightly curved towards the anteroapical angle, not reaching margin of the wing.

Fig. 10. Aleyrodidae gen. et sp. indet. 4 (NHMD 38963 / ZMUC 902136), Eocene, Danish amber. A1, A7, fore wing; A2, A6, hind wing; A3, metaleg; A4, body in ventral view; A5, body in dorsal view with wings; A8, antenna; A9, legs; A10, abdomen, wax plates. Explanatory drawings (A1–A3), photographs (A4–A10).
Prolegs the shortest, metalegs the longest (Fig. 10A3, A4, A9). Coxa elongated, trochanters poorly marked. Metatibia 1.4 times longer than metafemur. Metatibia and basimetatarsomere with small, short seta (Fig. 10A3, A4, A9, A10). Single, several times longer seta on the apical part of tibia. Metatarsus two-segmented, with basimetatarsomere 1.3 times longer than apical metatarsomere. The total length of metatarsus the same as length of metafemur. Paronychium and claws invisible.
Abdomen from the dorsal side invisible (Fig. 10A5). Ventral side of abdomen oval shape, similar in length to the head and thorax (Fig. 10A4, A10). Four pairs of wax plates visible, of similar size and regular shape, slightly wider than their length (Fig. 10A4, A10). Gender cannot be determined, probably female due to the characteristic triangular shape the apical part of the abdomen (visible from the lateral side) and lack of claspers and aedeagus (Fig. 10A4, A10).
Key to species of Gregorites Drohojowska & Szwedo gen. nov.
1. Vein CuA, branches Rs and CuA reaching margin of fore wing (Fig. 5A1); hind wing strongly narrowed basally and strongly wide apically (Fig. 5A2), almost all parts of body covered by very tiny, densely dispersed hairs ...................................................................................... Gregorites thrymi Drohojowska & Szwedo sp. nov.
– Vein CuA, branches R1, Rs and CuA not reaching margin of fore wing; hind wing not strongly narrowed basally; body not covered by hairs ................................................................................ 2
2. Fore wing with vein Rs arcuate, directed towards the posteroapical angle (Fig. 4A1); length of fore wing 1.2 mm ........................................... Gregorites bestlae Drohojowska & Szwedo sp. nov.
– Fore wing with vein Rs straight, length of fore wing more than 1.3 mm ................................................................................................... 3
3. Hind wing very short and broad (Fig. 2A2), mesopostnotum diamond-shaped, anterior margin strongly rounded about 1.2 times longer than its width (Fig. 2A6) ................................................................... Gregorites bergelmiri Drohojowska & Szwedo sp. nov.
– Hind wing in different shape, mesopostnotum triangular shaped, wider than long or in equal size ....................................................... 4
4. Fore wing with vein CuP longer than half of length of fore wing (Fig. 1A1) ............................................................................................................................ Gregorites aegiri Drohojowska & Szwedo sp. nov.
– Fore wing with vein CuP shorter than half of length of fore wing ................................................................................................................. 5
5. Compound eyes with anterior margin shallowly incised, ommatidia in upper and lower portion of compound eyes differentiated (Fig. 7A9) .................................................................................................................. Gregorites ymiri Drohojowska & Szwedo sp. nov.
– Compound eyes with anterior margin convex and not incised, ommatidia not differentiated .............................................................. 6
6. Fore wing gradually widening towards the apex (Fig. 6A1, A13), length of fore wing more 1.6 mm, width more 0.8 ......................................................... Gregorites halogii Drohojowska & Szwedo sp. nov.
– Fore wing more than 2 times as long as wide (Fig. 3A1), length of fore wing 1.38 mm, width 0.56 .................................................................................... Gregorites skadii Drohojowska & Szwedo sp. nov.
Discussion
The genus Gregorites Drohojowska & Szwedo gen. nov. and its species described above are the first whiteflies to be identified in Eocene amber from Denmark. The newly described genus could be placed in the subfamily Aleurodicinae, based on the venation pattern of the fore wing—with separate forking of Sc+R into R1 and Rs, structure of paronychium, a specific number of wax areas, and a distinctive shape of compound eyes observed from above. Fossil species of whiteflies described above characterized by the presence of seven antennomeres, a feature that is shared by the majority of modern representatives of the group. In contrast to the Recent species of whiteflies, the fossil species of the genus Gregorites Drohojowska & Szwedo gen. nov. do not exhibit a distinct division of the compound eyes, with the exception of Gregorites ymiri Drohojowska & Szwedo sp. nov., in which the anterior margin is shallowly incised but the ommatidia are not differentiated. In the case of extant species of Aleyrodidae, the compound eyes are often divided, with the upper and lower portions not connected or connected by single, two, three or four ommatidia; furthermore the ommatidia in the upper portion of the compound eye are different in size, typically larger, than those in the lower portion of the compound eye (Gerling 1990; Shahbazvar et al. 2011). The differences of Gregorites Drohojowska & Szwedo gen. nov. and other Aleurodicinae known from the Eocene ambers are the ratios of fore wing veins R1/Rs which measures 0.43 to 0.6 in Gregorites Drohojowska & Szwedo gen. nov., while in Paernis it measures 0.67 and in Rovnodicus it is 0.18. A similar disparity is observed among genera of Aleyrodidae from Oise amber, with values 0.53 in Oisedicus Drohojowska & Szwedo, 2013, 0.61 in Clodionus Drohojowska & Szwedo, 2013, 0.33 in Lukotekia Drohojowska & Szwedo, 2013, 0.38 in Isaraselis Drohojowska & Szwedo, 2013. Hind wing ratio R1/Rs in Gregorites Drohojowska & Szwedo gen. nov. is also variable, ranging from 0.3 to 0.56, while in Paernis it measures 1.
The taxonomic diversity of the Eocene Aleyrodidae preserved as inclusions in fossil resins (Fig. 11, Table 2) counts now ten genera (six from Baltic amber and four genera from Oise amber), with seventeen species (thirteen and four species, respectively). Within fossils from the Baltic amber group resins, the fossils from the Gulf of Gdańsk area include “Aleyrodes” aculeatus Menge, 1856, of unknown taxonomic status, two aleyrodine species, Snotra christelae Szwedo & Drohojowska, 2016, and Snotra herczeki Drohojowska & Szwedo, 2023, and two aleurodicine species, Paernis gregorius and Medocellodes blackmani. The amber of Rovno evidences the fauna of this area at this period of time which includes one aleurodicine species, Rovnodicus wojciechowskii. This study adds one more aleurodicine genus with seven species. The slightly older, aged 53 Ma the Oise amber evidences the fauna of this area at this period of time which includes four monospecific genera of Aleurodicinae Clodionus fizoli Drohojowska & Szwedo, 2013, Isaraselis cladiva Drohojowska & Szwedo, 2013, Lukotekia menae Drohojowska & Szwedo, 2013, and Oisedicus maginus Drohojowska & Szwedo, 2013 (Table 2). Nevertheless, the actual taxonomic palaeodiversity is challenging to ascertain due to a paucity of knowledge regarding the imaginal characteristics of the group and the interest in its morphological disparity presented by adults; the current classification is based on puparia (Martin 2003; Drohojowska and Szwedo 2011b; Szwedo and Drohojowska 2016). It is also important to consider the potential biases associated with the collection and preservation of specimens (Pike 1984; Szwedo and Sontag 2009; McCoy et al. 2018; Solórzano Kraemer et al. 2018). A total of over 100 whitefly specimens from Baltic amber from the Gulf of Gdańsk are known to exist in various collections. In contrast, whitefly collection from Rovno amber inclusions comprises less than 30 specimens, with only two specimens known from Bitterfeld amber at present. In total, the collection of amber from Danish amber comprises eleven specimens. The specimens in these collections vary considerably in their condition, from well-preserved specimens that can be formally described to severely damaged and hardly identifiable specimens. Although fossil adult Aleyrodidae are well represented in collections, they have been poorly researched (Drohojowska et al. 2015). The systematics of whiteflies is currently based almost solely on the puparial stage, and adults can be identified only rarely. The morphology and structure of the adults is still the largest area of ignorance concerning whitefly taxonomy The study of whiteflies on imagines is beset with a significant and convoluted historical background that has undoubtedly diverted the attention of researchers in other directions (Martin 2003; Drohojowska and Szwedo 2011b). This is exemplified by the analysis of relationships within the Aleyrodinae (Manzari and Quicke 2006), which was undertaken with the use of only a few adult characters. The general lack of descriptions of extant adult whiteflies makes the description of fossil taxa especially challenging. This is also one of the hindrances in studies of extinct whiteflies preserved in fossilised resins and as imprints in sedimentary deposits.
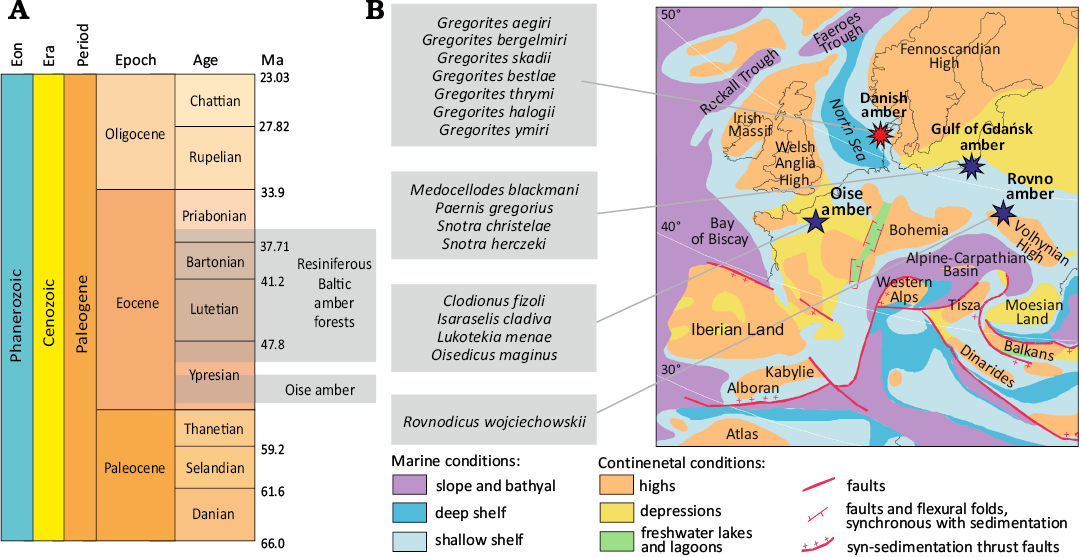
Fig. 11. Distribution of the Aleyrodidae in the Eocene fossil resins of Europe (asterisks). A. Chronostratigraphy after International Chronostratigraphic Chart 2023/09 (modified from Cohen et al. 2013). B. Palaeobiogeographic palinspastic background modified after Popov et al. (2009) and Iakovleva et al. (2020).
The fossil amber from the Paris Basin, Oise area is estimated to be ca. 53 Ma, Ypresian. It is deposited in beds comprising a succession of lenticular bodies, displaying two main facies: clayey sands rich in frequently pyritized lignite, accompanied by amber; and grey clayey sands with less lignite (1–12% of the sediment). These facies reflect a hypoxic environment, as evidenced by Brasero et al. (2009) and Nel and Brasero (2010). The formation of Oise amber coincided with a period of global warming, characterised by elevated temperatures and warm oceans (Gradstein et al. 2020; Inglis et al. 2020). This created a mild and humid environment, which enabled the “paratropical” forests to flourish in Europe (Kvaček 2010; Lenz et al. 2021). In Oise, the dominance of an arborescent amber-producing species and the presence of freshwater suggest a semi-deciduous forest (Nel et al. 2004). The producing amber tree is Aulacoxylon sparnacense (Combretaceae or Caesalpiniaceae) which could be close to the extant plants of the genus Terminalia (Combretaceae) or to Caesalpiniaceae (De Franceschi and De Ploëg 2003). It is noteworthy that Aleurodicinae are the predominant inclusions in this resin, with a single specimen of Aleyrodinae identified in the available samples to date. Species of Aleurodicinae are also more abundant and diverse than Aleyrodinae in mid- and late Eocene resins (amber from Denmark, Bitterfeld, Gulf of Gdańsk, and Rovno; Fig. 11; Table 2). The diversity of extant Aleyrodidae is also different within subfamilies. For example, there are 155 genera of Aleyrodinae (seventy two in the Oriental region to thirty in North America), and 21 genera of Aleurodicinae (fifteen in Neotropical region to one in the East Palaearctic). This is contrary to the pattern observed among fossils. To date only the Aleurodicinae have been identified in Danish amber. Similarly, the inclusions in amber from the Gulf of Gdańsk are dominated by Aleurodicinae, and a similar pattern is observed for the Rovno amber inclusions. During the Eocene several large-scale climatic and biotic changes occurred, resulting in a shift from a warm and equable climate to a seasonal and latitudinally stratified one. This was followed by a rapid global cooling at the Eocene–Oligocene Transition (Mosbrugger et al. 2005; Pound and Salzman 2017; Baatsen et al. 2020). The majority of extant whiteflies are thermophilous, with a nearly double number of species in the tropics compared to the temperate zone (Gerling 1990). This same biotic preference seems plausible to be assumed for the fossils, which could explain the diversity of these insects among inclusions in European Eocene resins. The observed differences in taxonomic composition among fossil sites may be attributed to a number of factors, including variations in the timing of amber formation, geographic and palaeofloristic differences, conditions in the palaeohabitats, and the taxic composition of palaeobiotas preserved in amber. In addition taphonomic circumstances and conditions must be considered (Pike 1984; Solórzano Kraemer et al. 2018; McCoy et al. 2018; Brysz et al. 2022). Conversely, observed differences may be attributed to past radiation events, which have resulted in diversification and disparification of whiteflies at local and regional scales (Nürk et al. 2020). The fossil resins collectively designate as “Baltic amber” are typically dated as Eocene, with a broad age range spanning from 50 to 37 million years ago (Ma). The difficulties in determining the age of Baltic amber are due to the repeated re-deposition of the amber, the extensive range of the ancient forest, and its probable existence for several million years. The hypotheses and opinions concerning the age of Baltic amber and its deposits were presented by Szwedo and Drohojowska (2016), Wolfe et al. (2016), Kosmowska-Ceranowicz (2017), Grimaldi and Ross (2017), Bogri et al. (2018), Szadziewski et al. (2018), Bukejs et al. (2019). The known deposits of Eocene amber probably arose from geographically (and chronologically?) independent sources (Larsson 1978; Wolfe et al. 2009, 2016; Mitov et al. 2021; Radchenko et al. 2021; Słodkowska et al. 2022). Currently, the deposits in Bitterfeld (Wolfe et al. 2016), Gulf of Gdańsk area (Szadziewski et al. 2018), Lublin area (Słodkowska et al. 2022), Rovno-Zhitomir region (Radchenko et al. 2021; Mitov et al. 2021) are typically regarded as distinct and independent fossil Lagerstätten. This also appears to be the case with amber from Denmark (Larsson 1978; Krause 1997; Shavrin et al. 2023). The recent discussions on the distinctive characteristics of amber and its inclusions, as well as the comparative analyses of the various groups of inclusions found in Baltic amber deposits from Rovno, the Gulf of Gdańsk, Bitterfeld and Denmark (Heie 1967, 1972; Heie and Wegierek 2011; Perkovsky 2016, 2017; 2018; Nadein et al. 2016; Simutnik et al. 2021; Shavrin et al. 2023) have led to the conclusion that the inclusive concept of “Baltic amber” (the Eocene succinite) found in the Baltic Sea Basin is misleading and should be abandoned. A more detailed understanding of the whiteflies found in these deposits could be employed in the recently proposed method of estimating the age of poorly dated fossil specimens and deposits using a total-evidence approach and the fossilized birth-death process (Barido-Sottani et al. 2023). It is evident that accurate dates for fossil specimens and deposits are of paramount importance, not only for elucidating the timing of speciation events in the tree of life, but also for providing crucial data for answering questions in evolutionary biology, paleoecology, biogeography, and paleoclimatology. Further investigations of morphological disparity, taxonomic diversity (extinct and extant) and palaeohabitat relationships of whiteflies are necessary to answer questions on evolutionary traits of these insects.
Table 2. List of Aleyrodidae species from Paleogene. * only formally placed in this genus. The taxon should be treated as incertae sedis within Aleyrodidae. Type material probably lost (Szwedo and Drohojowska 2016).
|
Taxon |
Reference |
Distribution |
Age |
|
Aleurodicinae |
|||
|
Clodionus |
|
|
|
|
Clodionus fizoli |
Oise, France |
Ypresian, lowermost Eocene |
|
|
Gregorites Drohojowska & Szwedo gen. nov. |
this paper |
|
|
|
Gregorites aegiri Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Gregorites bergelmiri Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Gregorites skadii Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Gregorites bestlae Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Gregorites thrymi Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Gregorites halogii Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Gregorites ymiri Drohojowska & Szwedo sp. nov. |
this paper |
succinite, Denmark |
Lutetian–Priabonian, Eocene |
|
Isaraselis |
|
|
|
|
Isaraselis cladiva |
Oise, France |
Ypresian, lowermost Eocene |
|
|
Lukotekia |
|
|
|
|
Lukotekia menae |
Oise, France |
Ypresian, lowermost Eocene |
|
|
Medocellodes |
|
|
|
|
Medocellodes blackmani |
Baltic amber, Gulf |
Lutetian–Priabonian, Eocene |
|
|
Oisedicus |
|
|
|
|
Oisedicus maginus |
Oise, France |
Ypresian, lowermost Eocene |
|
|
Paernis |
|
|
|
|
Paernis gregorius |
Baltic amber, Gulf |
Lutetian–Priabonian, Eocene |
|
|
Rovnodicus |
|
|
|
|
Rovnodicus wojciechowskii |
Baltic amber, Rovno deposit, Ukraine |
Lutetian–Priabonian, Eocene |
|
|
Aleyrodinae |
|||
|
Snotra |
|
|
|
|
Snotra christelae |
Baltic amber, Gulf |
Lutetian–Priabonian, Eocene |
|
|
Snotra herczeki |
Baltic amber, Gulf |
Lutetian–Priabonian, Eocene |
|
|
Aleyrodes |
Latreille 1796: 93 |
|
|
|
“Aleyrodes” aculeatus* |
Menge 1856: 18 |
Baltic amber |
Lutetian–Priabonian, Eocene |
Conclusions
We describe a new genus of whiteflies Gregorites Drohojowska & Szwedo gen. nov. with seven new species from the Eocene amber from Danish Coast, which represents subfamily Aleurodicinae. This subfamily is dominant in the Eocene fossil resins of Europe, contrary to modern fauna in the region which is dominated by Aleyrodinae. The new fossils shed a new light on taxonomic diversity and morphological disparity of the Aleyrodidae and rapid differentiation of these insects in the Eocene.
Acknowledgements
We wish to thank Lars Vilhelmsen ( Natural History Museum of Denmark in Copenhagen) for loaning the specimens. This research was funded in part by the National Science Centre, Poland, grant no. 2022/47/B/NZ8/02113 granted to JD. We are grateful to Gregory A. Evans (U.S. Department of Agriculture/Animal and Plant Health Inspection Service, USA) and an anonymous reviewer for constructive comments on an earlier draft of the manuscript.
Authors contribution
JD and JS designed research, wrote the paper and composed the final plates, generated the analyzed data and drafted taxonomic section, prepared final version of the manuscript; JŚ prepared photographs, drawings and morphological elaboration of the specimens.
References
Baatsen, M., Von Der Heydt, A.S., Huber, M., Kliphuis, M.A., Bijl, P.K., Sluijs, A., and Dijkstra, H.A. 2020. The middle to late Eocene greenhouse climate modelled using the CESM 1.0.5. Climate of the Past 16: 2573–2597.Crossref
Barido-Sottani, J., Żyła, D., and Heath, T.A. 2023. Estimating the age of poorly dated fossil specimens and deposits using a total-evidence approach and the fossilized birth-death process. Systematic Biology 72 (2): 466–475. Crossref
Bartlett, C.R., Deitz, L.L., Dmitriev, D.A., Sanborn, A.F., Soulier-Perkins, A., and Wallace, M.S. 2018. The diversity of the true hoppers (Hemiptera: Auchenorrhyncha). In: A.G. Foottit and P.H. Adler (eds.), Insect Biodiversity. Science and Society, Vol. 2, 501–590. John Wiley & Sons Ltd., Chichester. Crossref
Bogri, A., Solodovnikov, A., and Żyła, D. 2018. Baltic amber impact on historical biogeography and palaeoclimate research: Oriental rove beetle Dysanabatium found in the Eocene of Europe (Coleoptera, Staphylinidae, Paederinae). Papers in Palaeontology 4: 433–452. Crossref
Brasero, N., Nel, A., and Michez, D. 2009. Insects from the Early Eocene amber of Oise (France): diversity and palaeontological significance. Denisia 26: 41–52.
Brysz, A.M., Bourgoin, T., and Szwedo, J. 2022. Spotted beauty—Gedanochila museisucini gen. nov. et sp. nov.—a new Achilidae from Eocene Baltic amber (Hemiptera: Fulgoromorpha) and its relation to Achilini. Zootaxa 5125 (3): 295–312. Crossref
Bukejs, A., Alekseev, V.I., and Pollock, D.A. 2019. Waidelotinae, a new subfamily of Pyrochroidae (Coleoptera: Tenebrionoidea) from Baltic amber of the Sambian peninsula and the interpretation of Sambian amber stratigraphy, age and location. Zootaxa 4664 (2): 261–273. Crossref
Cohen, K.M., Finney, S.C., Gibbard, P.L., and Fan, J.-X. 2013 (updated). The ICS International Chronostratigraphic Chart. Episodes 36: 199–204. Crossref
De Franceschi, D. and De Ploëg, G. 2003. Origine de l’ambre des faciès sparnaciens (Eocène inférieur) du bassin de Paris: le bois de l’arbre producteur. Geodiversitas 25 (4): 663–647.
Drohojowska, J. and Szwedo, J. 2011a. A new whitefly from Lower Cretaceous Lebanese amber (Hemiptera: Sternorrhyncha: Aleyrodidae). Insect Systematics and Evolution 42 (2): 179–196. Crossref
Drohojowska, J. and Szwedo, J. 2011b. New Aleyrodidae (Hemiptera: Sternorrhyncha: Aleyrodomorpha) from the Eocene Baltic amber. Polish Journal of Entomology 80 (4): 659–677. Crossref
Drohojowska, J. and Szwedo, J. 2013a. The first Aleyrodidae from the Lowermost Eocene Oise amber (Hemiptera: Sternorrhyncha). Zootaxa 3636: 319–347. Crossref
Drohojowska, J. and Szwedo, J. 2013b. Gapenus rhinariatus gen. sp. n. from the Lower Cretaceous amber of Lebanon (Hemiptera: Sternorrhyncha: Aleyrodidae). In: D. Azar, M.S. Engel, E. Jarzembowski, L. Krogmann, A. Nel, and J. Santiago-Blay (eds.), Insect Evolution in an Amberiferous and Stone Alphabet. Proceedings of the 6th International Congress on Fossil Insects, Arthropods and Amber, 99–110. Brill, Leiden-Boston. Crossref
Drohojowska, J. and Szwedo, J. 2015. Early Cretaceous Aleyrodidae (Hemiptera: Sternorrhyncha) from the Lebanese amber. Cretaceous Research 52B: 368–389. Crossref
Drohojowska, J., Perkovsky, E.E., and Szwedo, J. 2015. New genus and species of Aleyrodidae from Eocene Baltic amber (Sternorrhyncha: Aleyrodomorpha. Polish Journal of Entomology 84: 259–269. Crossref
Drohojowska, J., Szwedo, J., Żyła, D., Huang, D.-Y., and Müller, P. 2020. Fossils reshape the Sternorrhyncha evolutionary tree (Insecta, Hemiptera). Scientific Reports 10: 11390. Crossref
Drohojowska, J., Tomanek, N., Gröhn, C., and Szwedo, J. 2022. A second aleurodicinae from the Eocene Baltic amber—Medocellodes blackmani gen. et sp. nov. (Hemiptera, Sternorrhyncha, Aleyrodidae). Zootaxa 5183: 245–253. Crossref
Drohojowska, J., Wegierek, P., Evans, G.A., and Huang, D. 2019. Are contemporary whiteflies “living fossils”? Morphology and systematic status of the oldest representatives of the Middle–Late Jurassic Aleyrodomorpha (Sternorrhyncha, Hemiptera) from Daohugou. Palaeoentomology 2: 171–182. Crossref
Dybkjær, K., Rasmussen, E.S., Eidvin, T., Grøsfjeld, K., Riis, F., Piasecki, S., and Śliwińska, K.K. 2020. A new stratigraphic framework for the Miocene–Lower Pliocene deposits offshore Scandinavia: A multiscale approach. Geological Journal 56: 1699–1725. Crossref
Evans, G.A., Martin, J.H., Drohojowska, J., Szwedo, J., Dubey, A.K., Dooley, J.W., and Stocks, I.C. (in press). Whiteflies of the World (Hemiptera: Sternorrhyncha, Aleyrodidae)—a catalogue of the taxonomy, distribution, hosts and natural enemies of whiteflies. Zootaxa.
Gerling, D. 1990. Whiteflies: Their Bionomics, Pest Status and Management. 348 pp. Intercept Ltd., Andover.
Gibbard P.L. and Lewin J. 2016. Filling the North Sea Basin: Cenozoic sediment sources and river styles. André Dumont medallist lecture 2014. Geologica Belgica 19: 201–217. Crossref
Gradstein, F.M., Ogg, J.G., Schmitz, M.D., and Ogg, G.M. 2020. Geologic Time Scale 2020. Elsevier, Amsterdam. Crossref
Grimaldi, D. and Engel, M.S. 2005. Evolution of the Insects. 772 pp. Cambridge University Press, Cambridge.
Grimaldi, D.A. and Ross, A.J. 2017. Extraordinary Lagerstätten in amber, with particular reference to Cretaceous of Burma. In: N. Fraser and H.D. Sues (eds.), Terrestrial Conservation Lagerstätten: Windows into the Evolution of Life on Land, 287–342. Dunedin Academic Press, Edinburgh. Crossref
Hakim, M., Azar, D., Szwedo, J., Drohojowska, J., and Huang, D.Y. 2021. Paraprotopsyllidiidae fam. nov., a new thrips-like protopsyllidioid family from mid-Cretaceous Burmese amber (Hemiptera; Sternorrhyncha). Cretaceous Research 120: 104726, 1–20. Crossref
Heie, O.E. 1967. Studies on fossil aphids (Homoptera: Aphidoidea). Spolia Zoologica Musei Hauniensis 26: 1–274.
Heie, O.E 1972. Some new fossil aphids from Baltic amber in the Copenhagen collection (Insecta, Homoptera: Aphididae). Steenstrupia 2: 247–262.
Heie, O.E. and Wegierek P. 2011. A list of fossil aphids (Hemiptera, Sternorrhyncha, Aphidomorpha). Monographs of the Upper Silesian Museum 6: 1–82.
Henry, T.J. 2017. Biodiversity of Heteroptera. In: A.G. Foottit and P.H. Adler (eds.), Insect Biodiversity. Science and Society, Vol. 1, 2nd Edition, 279–336. John Wiley & Sons Ltd., Chichester. Crossref
Iakovleva, A.I., Waga, D.D., and Andreeva-Grigorovich, A.S. 2020. New palynological data from the Middle Eocene deposits of the Kheu reference section (Kabardino-Balkaria, North Caucasus). Stratigraphy and Geological Correlation 28: 88–106. Crossref
Inglis, G.N., Bragg, F., Burls, N.J., Cramwinckel, M.J., Evans, D., Foster, G.L., Huber, M., Lunt, D.J., Siler, N., Steinig, S., Tierney, J.E., Wilkinson, R., Anagnostou, E., De Boer, A.M., Dunkley Jones, T., Edgar, K.M., Hollis, C.J., Hutchinson, D.K., and Pancost, R.D. 2020. Global mean surface temperature and climate sensitivity of the early Eocene Climatic Optimum (EECO), Paleocene–Eocene Thermal Maximum (PETM), and latest Paleocene. Climate of the Past 16: 1953–1968. Crossref
Klug, B. and Klug, G. 2015. Bernstein auf Fanø. 204 pp. Pro Business GmbH, Berlin.
Kosmowska-Ceranowicz, B. 2017. Amber in Poland and in the World. Second Edition, Revised. 310 pp. Wydawnictwa Uniwersytetu Warszawskiego, Warsaw.
Krause, K. 1997. Durch Gletscher und Wasser verfrachtet: Bernstein—Transport und Transportwege. Arbeitskreis Paläontologie Hannover 25: 166–174.
Kvaček, Z. 2010. Forest flora and vegetation of the European early Palaeogene—A review. Bulletin of Geosciences 85: 63–76. Crossref
Larsson, S.G. 1978. Baltic Amber: A Paleobiological Study (Entomograph). 192 pp. Scandinavian Science Press, Klampenborg. Crossref
Lenz, O.K., Riegel, W., and Wilde, V. 2021. Greenhouse conditions in lower Eocene coastal wetlands?—Lessons from Schöningen, Northern Germany. PLoS One 16 (1): e0232861. Crossref
Manzari, S. and Quicke, D.L.J. 2006. A cladistic analysis of whiteflies, subfamily Aleyrodinae (Hemiptera: Sternorrhyncha: Aleyrodidae). Journal of Natural History 44–46: 2423–2554. Crossref
Martin, J.H. 2003. Whiteflies (Hemiptera: Aleyrodidae)—their systematic history and the resulting problems of conventional taxonomy, with special reference to descriptions of Aleyrodes proletella (Linnaeus, 1758) and Bemisia tabaci (Gennadius, 1889). Entomologists’s Gazette 54: 125–136.
Martin, J.H. 2007. Giant whiteflies (Sternorrhyncha, Aleyrodidae): a discussion of their taxonomic and evolutionary significance, with the description of a new species of Udamoselis Enderlein from Ecuador. Tijdschrift voor Entomologie 150: 13–29. Crossref
McCoy, V.E., Soriano, C., Pegoraro, M., Luo, T., Boom, A., Foxman, B., and Gabbott, S.E. 2018. Unlocking preservation bias in the amber insect fossil record through experimental decay. PLoS One 13 (4): e0195482. Crossref
Mitov, P.G., Perkovsky, E.E., and Dunlop, J.A. 2021. Harvestmen (Arachnida: Opiliones) in Eocene Rovno amber (Ukraine). Zootaxa 4984: 043–072. Crossref
Mosbrugger, V., Utescher, T., and Dilcher, D.L. 2005. Cenozoic continental climatic evolution of Central Europe. Proceedings of the National Academy of Sciences of the United States of America 102: 14964–14969. Crossref
Nadein, K.S., Perkovsky, E.E., Moseykonel, A.G., and Labandeira, C.C. 2016. New late Eocene chrysomelidae (Insecta: Coleoptera) from Baltic, Rovno and Danish ambers. Papers in Palaeontology 2: 117–137.
Nel, A. and Brasero, N. 2010. Oise amber. In: D. Penney (ed.), Biodiversity of Fossils in Amber From the Major World Deposits, 137−148. Siri Scientific Press, Manchester.
Nel, A., De Ploëg, G., Millet, J., Menier, J.-J., and Waller, A. 2004. The French ambers: a general conspectus and the Lowermost Eocene amber deposit of Le Quesnoy in the Paris Basin. Geologica Acta 2: 3–8.
Nürk, N.M., Linder, H.P., Onstein, R.E., Larcombe, M.J., Hughes, C.E., Piñeiro Fernández, L., Schlüter, P.M., Valente, L., Beierkuhnlein, C., Cutts, V., Donoghue, M.J., Edwards, E.J., Field, R., Flantua, S., Higgins, S.I., Jentsch, A., Liede-Schumann, S., and Pirie, M.D. 2020. Diversification in evolutionary arenas—assessment and synthesis. Ecology and Evolution 10: 6163–6182. Crossref
Pedersen, G.K. and Pedersen, S.A.S. 2013. Geology of the early Eocene Fur Formation, a unique deposit in the North Sea Basin. Excursion guide to geological key localities on Mors and Fur, northern Denmark. Prepared for Wintershall Norge AS. GEUS. Danmarks og Grønlands Geologiske Undersøgelse Rapport 47: 1–55.
Perkovsky, E.E. 2016. Tropical and Holarctic ants in late Eocene ambers. Vestnik zoologii 50: 111–122. Crossref
Perkovsky, E.E. 2017. Comparison of biting midges of the early Eocene Cambay amber (India) and late Eocene European ambers supports the independent origin of European ambers. Vestnik zoologii 51: 275–284. Crossref
Perkovsky, E.E. 2018. Only a half of species of Hymenoptera in Rovno amber fauna is common with Baltic amber. Vestnik zoologii 52: 353–360. Crossref
Pike, E.M. 1984. Amber taphonomy and collecting biases. Palaios 8 (5): 411–419. Crossref
Poinar, G.O., Jr. 1992. Life in Amber. 368 pp. Stanford University Press, Stanford. Crossref
Popov, S.V., Akhmetiev, M.A., Lopatin, A.V., Bugrova, E.M., Sychevskaya, E.K., Shcherba, I.G., Andreeva-Grigorovich, A.S. , Zaporozhets, N.I., Nikolaeva, I.A., and Kopp, M.L. 2009. Paleogeografiâ i biogeografiâ basseinov Paratetisa. Čast’ 1. Pozdnij eocen-rannyj miocen. Naučnyj Mir, Moskva.
Pound, M. and Salzmann, U. 2017. Heterogeneity in global vegetation and terrestrial climate change during the late Eocene to early Oligocene transition. Scientific Reports 7: 43386. Crossref
Radchenko, A.G., Perkovsky, E.E., and Vasiliev, D.V. 2021. Formica species (Hymenoptera, Formicidae, Formicinae) in late Eocene Rovno amber. Journal of Hymenoptera Research 82: 237–251. Crossref
Rietschel, S. 1983. Aleurochiton petri n. sp., eine Mottenschildlaus (Homoptera, Aleyrodina) aus dem Pliozän von Neu-Isenburg, Hessen. Carolinea 41: 97−100.
Schlee, D. 1970. Verwandtschaftsforschung an fossilen und rezenten Aleyrodina (Insecta, Hemiptera). Stuttgarter Beiträge zur Naturkunde 213: 1–72.
Schmidt, A.R., Perrichot, V., Svojtka, M., Anderson, K.B., Belete, K.H., Bussert, R., Dörfelt, H., Jancke, S., Mohr, B., Mohrmann, E., Nascimbene, P.C., Nel, A., Nel, P., Ragazzi, E., Roghi, G., Saupe, E.E., Schmidt, K., Schneider, H., Selden, P.A., and Vávra, N. 2010. Cretaceous African life captured in amber. Proceedings of the National Academy of Sciences of the United States of America 107: 7329−7334. Crossref
Shahbazvar, N., Sahragard, A., Hosseini, R., and Hajizadeh, J. 2011. A preliminarily study on adult characters of whiteflies (Hem.: Aleyrodidae). Entomofauna. Zeitschrift für Entomologie 32: 413–420.
Shavrin, A.V., Solodovnikov, A., Kairišs, K., and Jenkins Shaw, J. 2023. A new Acrulia species from Danish amber (Coleoptera: Staphylinidae: Omaliinae: Omaliini). Historical Biology: 1–6. https://doi.org/10.1080/08912963.2023.2294348 Crossref
Shcherbakov, D.E. 2000. The most primitive whiteflies (Hemiptera; Aleyrodidae; Bernaeinae subfam. nov.) from the Mesozoic of Asia and Burmese amber, with an overview of Burmese amber hemipterans. Bulletin of the Natural History Museum, Geology Series 56: 29–37.
Simonsen, S. 2008. Bernsteinsuche auf der Insel Fanø. Arbeitskreis Paläontologie Hannover 36: 122–128.
Simutnik, S.A., Perkovsky, E.E., Khomych, M.R., and Vasilenko, D.V. 2021. First record of the Sulia glaesaria Simutnik, 2015 (Hymenoptera, Chalcidoidea, Encyrtidae) from Rovno amber. Journal of Hymenoptera Research 88: 85–102. Crossref
Słodkowska, B., Kasiński, J.R., and Żarski, M. 2022. Stratigraphic and environmental conditions of the occurrence of amber-bearing deposits in the northern Lublin region. Przegląd Geologiczny 70: 50–60.
Solórzano Kraemer, M.M., Delclòs, X., Clapham, M.E., Arillo, A., Peris, D., Jäger, P., Stebner, F., and Peñalver, E. 2018. Arthropods in modern resins reveal if amber accurately recorded forest arthropod communities. Proceedings of the National Academy of Sciences of the United States of America 115: 6739–6744. Crossref
Szadziewski, R., Szwedo, J., and Sontag, E. 2018. Fauna lasu bursztynowego / Fauna of the amber forest. In: R. Szadziewski, R. Pytlos, and J. Szwedo (eds.), Bursztyn bałtycki – skarb Zatoki Gdańskiej / Baltic amber – treasure of the Bay of Gdańsk, 38–75, 216–217. Związek Miast i Gmin Morskich, Gdańsk.
Szwedo, J. 2018. The unity, diversity and conformity of bugs (Hemiptera) through time. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 107: 109–128. Crossref
Szwedo, J. and Drohojowska, J. 2016. A swarm of whiteflies—the first record of gregarious behavior from Eocene Baltic amber. The Science of Nature 103 (35): 1–6 + 1–26. Crossref
Szwedo, J. and Sontag, E. 2009. The traps of the “amber trap”. Amber-trapped insects trap scientists with enigmas. In: B. Berning. and S. Podenas (eds.), Amber: Archive of the Deep Time. Denisia, 26, zugleich Kataloge der oberösterreichischen Landesmuseen Neue Serie 86: 155–169. Biologiezentrum der Oberösterreichischen Landesmuseen, Linz.
Szwedo, J., Drohojowska, J., Popov, Yu.A., Simon, E., and Wegierek, P. 2019. Aphids, true hoppers, jumping plant-lice, scale insects, true bugs and whiteflies (Insecta: Hemiptera) from the Insect Limestone (latest Eocene) of the Isle of Wight, UK. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 110 (3–4): 331–396. Crossref
Weber, H. 1935. Der Bau der Imago der Aleurodinen. Ein Beitrag zur vergleichenden Morphologie des Insektenkorpers. Zoologica 33, Band 8 (Heft 89): 1–71.
Wolfe, A.P., Mckellar, R.C., Tappert, R., Sodhi, R.N.S., and Muehlenbachs, K. 2016. Bitterfeld amber is not Baltic amber: Three geochemical tests and further constraints on the botanical affinities of succinite. Review of Palaeobotany and Palynology 225: 21–32. Crossref
Wolfe, A.P., Tappert, R., Muehlenbachs, K., Boudreau, M., Mckellar, R.C., Basinger, J.F., and Garrett, A. 2009. A new proposal concerning the botanical origin of Baltic amber. Proceedings of the Royal Society B 276: 3403–3412. Crossref
Acta Palaeontol. Pol. 69 (4): 649–676, 2024
https://doi.org/10.4202/app.01173.2024