

Predator-predator-prey interaction between spiders and insects: First fossil evidence from 23 million-year old Chiapas amber syninclusion
MIGUEL ÁNGEL GARCÍA-VILLAFUERTE and GERARDO CARBOT-CHANONA
García-Villafuerte, M.A. and Carbot-Chanona, G. 2024. Predator-predator-prey interaction between spiders and insects: First fossil evidence from 23 million-year old Chiapas amber syninclusion. Acta Palaeontologica Polonica 69 (4): 677–685.
Syninclusions are palaeontological resources that provide palaeoautoecological evidence of fossil species and information on the biological interactions between different organisms that were part of a past ecosystem. Although palaeautoecological interactions in amber have been documented worldwide, interactions between predators and potential prey are rare. Here, we documented the first evidence in Miocene Chiapas amber of predator-predator-prey interaction involving two spider species and one insect: the araneophagous “pirate spider” Mimetus sp., the Theridiidae spider Thymoites carboti, and gall flies (Cecydomiidae). The interaction between Mimetus sp. and T. carboti is documented as a possible case of araneophagy or opportunism. Also, the first evidence of a web built by some members of the Thymoites genus is presented. The taphonomic analysis of the amber piece indicated that they were all captured at the same time under the same resin flow.
Key words: Early Miocene, araneophagy, biological interactions, palaeoautoecology, syninclusions, Chiapas, Mexico.
Miguel Ángel García-Villafuerte [mgarciavillafuerte@yahoo.com.mx; ORCID: https://orcid.org/0000-0003-0163-0736 ], Independent Researcher. Tuxtla Gutiérrez, Chiapas, México.
Gerardo Carbot-Chanona [gfcarbot@gmail.com; ORCID: https://orcid.org/0000-0001-9582-9273 ] (corresponding author), Museo de Paleontología “Eliseo Palacios Aguilera”, Departamento de Paleontología, Dirección de Gestión, Investigación y Educación Ambiental, Secretaría de Medio Ambiente e Historia Natural. Calzada de Las Personas Ilustres, s/n, 29000, Tuxtla Gutiérrez, Chiapas, Mexico.
Received 23 July 2024, accepted 9 October 2024, published online 19 December 2024.
Copyright © 2024 M.Á. García-Villafuerte and G. Carbot-Chanona. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Preservation of organisms in amber is an excellent source of biological information, particularly on invertebrates (e.g., Solórzano-Kraemer and Brown 2017; García-Villafuerte 2018a). The organisms trapped in the resin usually die so quickly that they could have been fossilized in real-life time lapse (Arillo 2007). In this way, it is feasible to analyse the possible biological interactions between different inclusions preserved in the same piece of amber and their possible relationship with the environment in which they lived. Arillo (2007) mentioned that when two or more organisms are embedded in the same resin flow, they probably lived and died on the same day; therefore, there is a possibility that these organisms lived in the same ecosystem. Associations between two or more organisms embedded in the same amber piece are called syninclusions (Koteja 1996).
Palaeoecological inferences are based on the principle of biological actualism, which dictates that phenomena observed today can be studied in the past (Cevallos-Ferriz and Ramírez 1998). Based on the previous premise, two methods have been proposed to infer the lifestyle of fossil species: (i) morphofunctional studies, also known as ecomorphological studies, which analyse how the shape of fossil remains allowed, restricted or caused the functions that an organism could perform in its habitat (Plotnick and Baumiller 2000; González-Guarda 2019; Vizcaíno et al. 2006); and (ii) the implementation of taxonomic analysis, which means that the lifestyle of extinct species is assumed based on the behaviour of the extant species with which they are more closely related (Dood and Stanton 1981). However, when it comes to the behaviour or ethology of living beings, the processes can be more complicated to apply in palaeoecology because individuals, when adapting to new conditions, may not maintain the same behaviour. Therefore, it is important to exercise caution with this interpretation in palaeontological and palaeobiological studies (Cevallos-Ferriz and Ramírez 1998).
Syninclusions are palaeontological resources that provide palaeoautoecological evidence of fossil species and information on the biological interactions between different organisms that were part of a past ecosystem (Arillo 2007). For example, cases of parasitism in insects preserved in amber from Spain, the Dominican Republic, and the Baltic have been recorded (Poinar and Miller 2002; Arillo 2007; Belokobylskij et al. 2021). Similarly, there are fossil records of spiders parasitized by fungi, nematodes, mites, and insect larvae from Baltic and Dominican amber (Wunderlich 2004), as well as termites parasitized by Sordariomycetes fungi from Chiapas amber (Arroyo-Sánchez et al. 2023).
Although autoecological interactions in amber have been documented worldwide, interactions between predators and potential prey are rare. The spider Geratonephila burmanica Poinar, 2012 (Nephilidae) preying on the platygastrid wasp Cascoscelio incassus Poinar, 2012 (Poinar and Buckley 2012), a worker ant of Ceratomyrmex ellenbergeri Perrichot, Wang & Engel, 2016, grasping the alienopteran nymph of Caputoraptor elegans Bai, Beutel & Wipfle, 2018 (Barden et al. 2020), and indirect evidence of a spider preying on ticks (Dunlop et al. 2018; de la Fuente et al. 2024), have been documented in mid-Cretaceous Burmese amber. Some cases of araneophagy (Wunderlich 2004, 2008), also predator-prey interactions between the zodariid spider Anniculus balticus Petrunkevitch, 1942, and an ant (Penney and Selden 2011), and lacewing larvae (Neuroptera) with possible aphid prey (Haug et al. 2023) have been documented in the Baltic amber. From the Chiapas amber there are reports of a theridiid spider Theridion Walckenaer, 1805, preying on a dolichopodid fly (García-Villafuerte 2018a), and a clubonid spider Prosocer Petrunkevitch, 1963, preying on an ant Azteca Forel, 1878 (Cordova-Tabares et al. 2024). Nevertheless, the three-way interaction between one predator and another predator and its prey has never been documented in amber.
In this study, we documented predator-prey (spider-insect) and predator-predator (spider-spider) interactions in a piece of amber from the Early Miocene Chiapas amber, that involving two spider species, theridiid Thymoites carboti García-Villafuerte, 2022, as predator and the mimetid pirate spider Mimetus sp. (García-Villafuerte 2022), with a possible case of araneophagy or opportunism. Additionally, a taphonomic interpretation of the studied amber piece is presented.
Institutional abbreviations.—IHNFG, Instituto de Historia Natural, Fósil Geográfico (historically is the acronym used for the Paleontological Collection, currently under the custody of the Palaeontology Museum “Eliseo Palacios Aguilera”, Secretaría de Medio Ambiente e Historia Natural (SEMAHN), Chiapas, Mexico.
Other abbreviations.—ant, antenna; cw, remains of cobweb; esp, spinnerets; gd, glue droplets; MB1, first level mounting band; MB2, second level mounting band; MB3, third level mounting band; Op, opisthosoma; rs, raptor spines; wg, wing remains.
Material and methods
The material studied comes from the Los Pocitos mines excavating in the Mazantic Shale that crop out in the municipality of Simojovel de Allende, State of Chiapas, southern Mexico (Fig. 1). The Mazantic Shale is formed by a sequence of 310 m of dark gray shales that contain amber in association with pollen, benthic foraminiferans, corals, echinoids, marine bivalves and gastropods, crabs, chondrichthyan remains, and terrestrial vertebrates (Langenheim et al. 1966; Frost and Langenheim 1974; Webb et al. 2003; Vega et al. 2009; Perrilliat et al. 2010; Carbot-Chanona et al. 2020). This lithological sequence has been dated with an absolute age of 23 Ma based on 87Sr/86Sr isotopes of a sample taken from a well-preserved shell of the gastropod Turbinella maya Perrilliat, Vega & Coutiño, 2010, placing it in the Aquitanian, Early Miocene (Vega et al. 2009).
Fig. 1. Location of Los Pocitos mines locality, Simojovel de Allende, State of Chiapas, southern Mexico, where the studied Early Miocene amber piece was obtained.
Originally, the studied piece had an oval shape, with the following dimensions: 30 mm wide, 18 mm long, and 15 mm thick. The amber piece was cut in half. In one half, there are only a few plant fragments, and this part was conserved. In the second half, there are all the inclusions described here; this section was cut into seven different fragments to facilitate taxonomic identification of the spider specimens and to properly identify the accompanying inclusions. Unfortunately, a photograph of the original piece was not taken, but a reconstruction was made, taking as reference the conserved sections of the original piece and using photographs of the preserved inclusions (Fig. 2). A catalog number was assigned to each fragment, as follow: male of Thymoites carboti IHNFG-5805; female of T. carboti IHNFG-5806; female of Mimetus sp. IHNFG-5808; Cedusa sp. (Derbidae) IHNFG-5809; small fruit IHNFG-5810; spider web remains IHNFG-5811; small piece with lines of formation IHNFG-6184.
The amber pieces were cut, sanded, and polished in the laboratory of the museum. A jeweler’s saw blade, adapted with a No. 1 blade, was used to cut. Subsequently, each piece was ground with different sandpaper grades (No. 320–No. 1500). Using a liquid abrasive, each piece was polished to reveal each specimen’s morphological characteristics (García-Villafuerte 2018b).
Information was collected using a Zeiss Stemi 2000-C stereomicroscope. Photographs were obtained using a digital camera attached to the stereomicroscope. Images were stacked using Helicon Focus software. Illustrations were created using Adobe Illustrator CC 2015 software. All measurements are given in millimeters and were taken with an ocular micrometer attached to the stereomicroscope. Layers present in the amber piece which were originated as a consequence of independent flows of resin in different time intervals, were called “assembly bands”.
Considering the thickness of the original piece of amber, as well as the internal hue, three areas were denominated “mounting bands” and each was designed as: MB1, MB2, and MB3, with the aim of delimiting the position of the specimens studied.

Fig. 2. General overview of the syninclusions, showing the relative position of female Mimetus sp. (IHNFG-5808), the female (IHNFG-5806), and male (IHNFG-5805) of Thyomites carboti; the web line of T. carboti (IHNFG-5811), the small fruit (IHNFG-5810), bees (not numbered), and Cedusa sp. (IHNFG-5809), from Early Miocene Chiapas amber, Mexico. Scale bars 500 µm.
Results
A total of 12 inclusions were identified in the piece of amber. In the MB3, which corresponded to the center of the piece, a planthopper of the genus Cedusa Fowler, 1904, (Hemiptera: Auchenorrhyncha: Derbidae) is present (Fig. 2). In MB2, meliponin bees (Hymenoptera: Apidae: Meliponini) are located at the edge of the original piece. In MB1, a female spider belonging to the genus Mimetus (Fig. 2), a lepidopteran larva, unidentified bryophytes, male and female Thymoites carboti spiders, three gall flies (Cecidomyiidae), unidentified insect remains, and small unidentified fruit were present (Fig. 2). The first gall fly is close to the female T. carboti mouthparts (Fig. 3A). Although this region is not clearly observable, it is evident that the fly is trapped between the chelicerae. The second fly is trapped between right leg I and left legs I and II of the same spider (Fig. 3A). Also, there are cobweb remains attached from the right legs I and II of T. carboti to this fly (Fig. 3B). Another fly is on the right side of this spider and has remains of spider web attached to it, which makes us to hypothesize that this fly was a previously hunted prey stored in the spider web. Another web line extending 18 mm in length is attached to leg II of the Mimetus sp. (Fig. 3C). Small glue droplets are also observed in the web lines attached to T. carboti and flies as well as in the lines connected to leg II of Mimetus sp. (Fig. 3B, C).
Each inclusion observed in the study piece present abnormal positions and a certain orientation in their morphological structures owing to resin flow, i.e., abnormal orientation in wing position, legs, chelicerae, pedipalps and antennae.
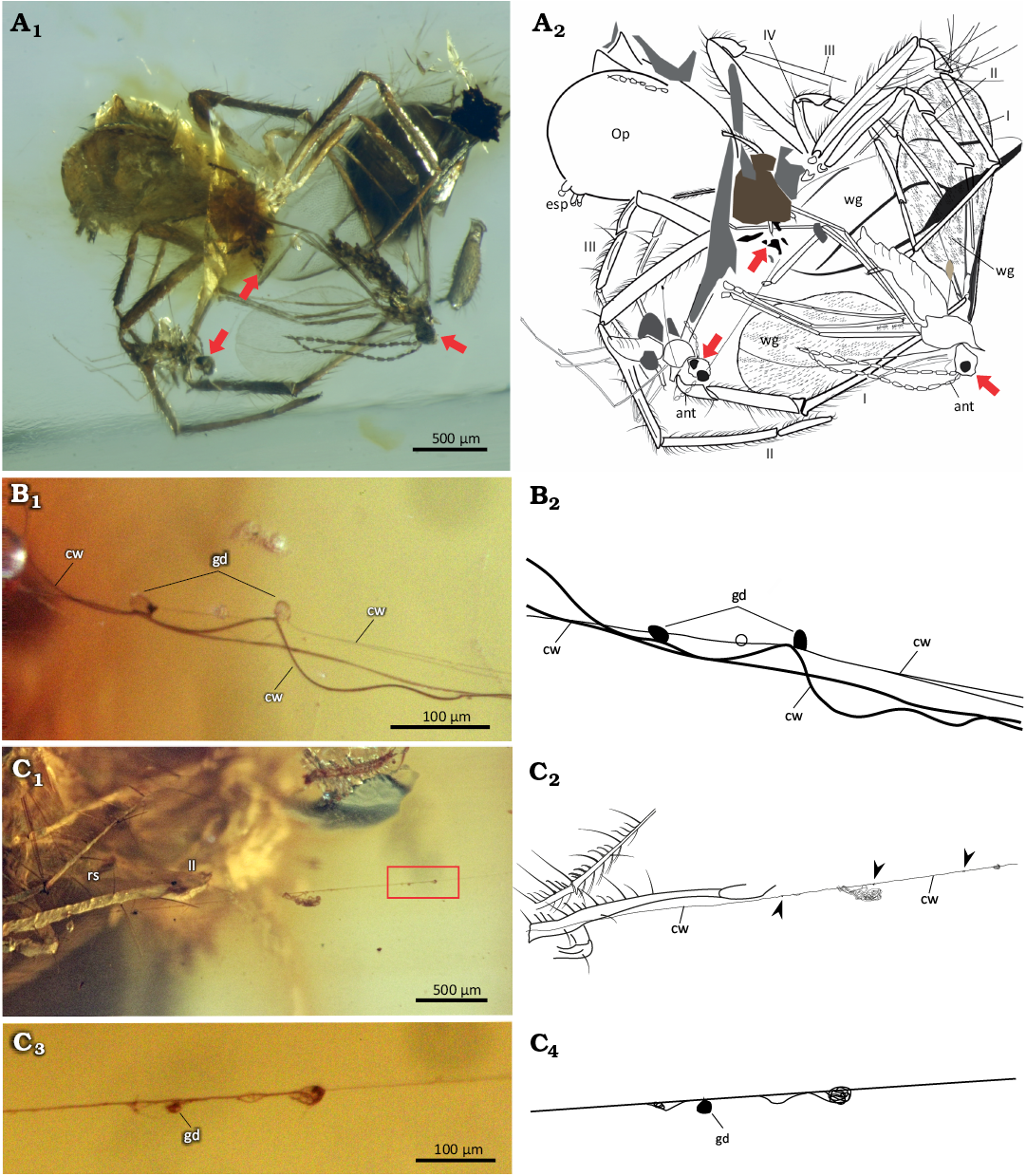
Fig. 3. Predator-predator-prey interactions recorded in an Early Miocene amber piece from Los Pocitos mines, Simojovel de Allende, Chiapas, Mexico. A. IHNFG-5806, the interaction between the female theridiid spider Thymoites carboti García-Villafuerte, 2022, and three gall flies (red arrows). B. IHNFG-5811, the cobweb of T. carboti showing the glue drops. C. IHNFG-5808, the interaction between the pirate spider Mimetus sp. and the cobweb of T. carboti (C1–C4); the detail (red rectangle in C1) of the cobweb attached to leg II of Mimetus sp. showing the glue droplet in the web line (arrowheads in C2). Roman numbers indicate the position of legs. Abbreviations: ant, antenna; cw, remains of cobweb; esp, spinnerets; gd, glue droplets; Op, opisthosoma; rs, raptor spines; wg, wing remains. Photographs (A1, B1, C1, C3) and schematic drawings (A2, B2, C2, C4). The position of syninclusions are showed in the Fig. 2.
Discussion
Spider-insect interaction.—The position of one gall fly trapped in the chelicerae and other between the first pair of legs of female Thymoites carboti, provides clear evidence that the spider was preying on these insects. Additionally, there are web lines joined to the legs I and II of the T. carboti and to the long legs and bodies of the three gall flies. These web lines have small glue droplets separated from each other along the web line (Fig. 3B), which is typical of the cobwebs used for prey capture by theridiid spiders. The web is attributed to T. carboti because Mimetus spiders do not build capture cobwebs (Benavides and Hormiga 2020). Although these interpretations should be taken with caution because sometimes it is possible to misinterpreted the predatory behavior when animals are found together (see Penney 2014; Poinar and Buckley 2014), based on the above observations presented here, we deduce that T. carboti captured gall flies as prey. This is the second record that involves Theridiidae (previously known from Theridium cf. hispidum) preying on these insects in Chiapas amber (García-Villafuerte 2018a). Although Cordova-Tabares et al. (2024) argued that such evidence is merely a speculative assumption, these authors did not examine the relative piece first-hand, which draws into question the validity of their criticism.
Members of the Theridiidae family present a wide range of spider webs and hunting strategies (Santos-González 2005). Many authors have described theridiid cobwebs built (e.g., Benjamin and Zschokke 2003; Eberhard et al. 2008); however, there is only one report of the cobweb structure of extant Thymoites pallidus (Emerton, 1913) (Guarisco 2000). Therefore, it is not well known whether the cobwebs of this genus have lines with glue droplets, whether the silk is viscous, whether they have a leaf-like web structure, or whether they possess modifications such as a retraction area or rest for the spider (e.g., Benjamin and Zschokke 2003; Eberhard et al. 2008). In the amber pieces IHNFG-5808 and IHNFG-5811, small glue droplets are separately distributed from each other along two lines which was once the T. carboti web line (Fig. 3B). The distribution of these glue droplets along the silk threads has an impact on prey retention in such a way, that threads with small glue droplets work with weak flying prey and long-legged insects, such as in the case of some groups of Diptera (Zschokke 2004; Eberhard et al. 2008). Based on the above, we hypothesize that it is likely that extant species of Thymoites build webs where silk lines contain small glue droplets spaced apart, which attach to the available surfaces, like other theridids (e.g., Zschokke 2003; Blackledge et al. 2005). However, this idea should be tested in the field or laboratory in living species of Thymoites. Therefore, here we documented the first cobweb evidence for Thymoites with the presence of small glue droplets (Fig. 3B, C).
Spider-spider interaction.—All spiders are predators although few species eat pollen and some species resort to araneophagy. This last practice is sometimes opportunistic in nature (Foelix 1982). However, there are some species which specialize in the capture and predation on spiders (Foelix 1982; Jackson and Hallas 1986; Jackson 1992; Pekár et al. 2024). Mimetids are generally presumed to be free-living obligate araneophages (Penney et al. 2012). These spiders are well known for their predatory tactics, invading webs, and using a vibrating lure to attract resident spiders within their deadly range (Kloock 2012). They even make movements on the nets of some theridids, as in the case of pirate spider Ero furcata (Villers, 1789), which forces the theridiid male to move away when he courts the female with the aim of taking his place and later slowly advancing towards the female. While this happens, it uses its front legs to hit the threads, until it finally makes a deadly attack (Czajka 1963). There have also been documented cases of opportunistic mimetids feeding on insects trapped in the webs of other spiders (Jackson and Whitehouse 1986).
Members of the genus Mimetus are araneophages of orbicular web-weaving species (e.g., Araneidae and Tetragnathidae), and irregular web-weaving spiders (e.g., Dictynidae and Theridiidae), as well as spiders that generally do not build webs (Philodromidae, Salticidae, and Thomisidae) (Jackson and Whitehouse 1986; Kloock 2001, 2012; Romero and Flórez 2014).
In the assembly band MB1, both females Mimetus sp., female T. carboti, and the gall flies occur, indicating that they were all captured at the same time under the same resin flow. In this process, one of the web lines of the T. carboti was in contact with the leg II of Mimetus sp. (Fig. 3C), suggesting that there may be some type of interaction between both spiders, either a possible case of araneophagy or opportunism. Although there are cases of Mimetus araneophagy reported in some theridiid species (e.g., Jackson and Whitehouse 1986; Kloock 2001, 2012), a case of specific interaction between extinct or extant members of Mimetus and Thymoites has not currently been recorded. Therefore, we document the first putative case of araneophagy of Mimetus on Thymoites.
Taphonomic notes.—When the resin is exuded, it may form structures in a shape of stalactites or drops (Weitschat and Wichard 1998). In this way, amber pieces are the result of a series of resin fluids originating from the tree species (Martínez-Delclòs et al. 2004). These fluids become flow lines, which are sometimes partially visible on the surface of the pieces but can be quite visible when the piece is cut and exposed to certain light orientations (Fig. 4). Internally, amber pieces can present these flow lines as sinuous lines (Martínez-Delclòs et al. 2004). In the original amber piece, 36 sinuous lines were observed running around it, forming several deposition circles (Fig. 4). This indicates that the descent of the resin was a vertical and constant flow. It is ruled out that these are internal fractures of the amber based on two arguments: (i) internal amber fractures usually originated during the biostratonomy and/or diagenesis process, and they do not have the appearance of smooth lines that run throughout the piece; (ii) fractures never follow the contour of inclusions; on the contrary, they often damage them (Coty et al. 2014). This is observed with the pirate spider included in piece IHNFG-5808, which is severely damaged due to its internal fractures.

Fig. 4. Details of an Early Miocene amber piece (IHNFG-6184) from Los Pocitos mines, Simojovel de Allende, Chiapas, Mexico, under indirect light where the lines of formation (black arrowheads) and the “mounting bands” (MB) are observed.
The observed evidence indicates that the resin production that led to the preservation of the piece analysed in this study, was continuous and fluid (see details on resin production and polymerization in Martínez-Delclòs et al. 2004), leaving thin layers, what is inferred from the short distance between each of the sinuous lines (Fig. 4). Likewise, according to Martínez-Delclòs et al. (2004), when we observe twisted wings, abnormal positions, or the specimens show oriented body structures (legs, antennae, wings, etc.), they generally reflect the constant flow of resin. This happens with the inclusions studied, in which abnormal positions and oriented body structures were observed. In contrast, drop-type amber pieces, disarticulate the limbs of the trapped organisms (Martínez-Delclòs et al. 2004).
Due the position of the planthopper of the genus Cedusa located closer to the center of the original piece (Fig. 2), it is possible that it was the first organism trapped by the resin. Weitschat and Wichard (1998) mentioned that amber occasionally preserves alternating light and dark bands, which represent successive flows. Martínez-Delclòs et al. (2004) pointed out that the darkest layers are generally the result of rapid drying by sunlight and the wind. Therefore, assembly band MB3 (Fig. 4) was produced by the first resin flows secreted by the tree, and it came into contact with Cedusa sp. Subsequent flows of resin formed the MB2, and the bees were trapped. Finally, resin descended and trapped the female Mimetus sp., the female of T. carboti, and the insects, forming the MB1. Probably this process took place at the base of the trunk of the tree that produced the resin, where sunlight and wind had less impact. Due to the presence of a small fruit (Fig. 2), we hypothesized that this occurred very close to the ground level, since the fruit is part of MB1.
Conclusions
Palaeoautoecological investigations of fossil spiders in Mexican amber are scarce. However, this type of research can provide important biological information that has not yet been recorded for extant representatives. This is the case provided by the spider web remains of Thymoites carboti, because until now, there has been no information on webs built by the extant species of Thymoites. Based on the palaeontological information presented here, we hypothesize that members of this genus build webs, with small drops of glue distributed along the capture lines of the web, making easier to capture prey with long legs and weak flight. However, the evidence presented here is based on a single individual, therefore pending more materials to get more conclusive results. Further research on this type of the web built by living Thymoites spiders is also required.
Predation by T. carboti on gall flies (Cecydomyiidae) in this study is evident since the insects are in contact with the web and one of them with the spider itself. On the other hand, although cases of araneophagy of extant members of Mimetus have been recorded in some species of Theridiidae, the interaction between extant species of Mimetus and Thymoites has never been documented. Although only one of the sticky silk lines of T. carboti was observed in contact with leg II of Mimetus sp., we suggest that, in the original piece, both spiders and gall flies were fossilized in a relatively small space, and the position as well as the alignment of the two species of spiders in this piece of amber could not be only fortuitous. Thus, an interaction between the two species probably occurred. Therefore, a possible case of araneophagy or opportunism, as well as the first evidence in amber of predator-predator-prey interaction preserved in Miocene amber from Chiapas, are documented.
Acknowledgements
We thank the Instituto de Ciencia, Tecnología e Innovación for the support granted for the study of biological inclusions in amber from Chiapas through the “Estudio de la araneofauna (Arachnida: Araneae) fósil y actual de Simojovel de Allende, Chiapas, México” and “Fortalecimiento del estudio de los microinvertebrados y microvertebrados fósiles del Estado de Chiapas, México, a través de la microfotografía digital” projects. We thank Luis Enrique Gómez-Pérez (Museo de Paleontología “Eliseo Palacios Aguilera”, Chiapas, Mexico) for the photographs and Paul Selden (Department of Geology, University of Kansas, Lawrence, USA) for their comments on an early draft of this manuscript. MAG-V appreciates the support of Gregorio García Miranda (Zacatecas, Mexico), for the continuity of this investigation. We also thank to Jason Dunlop (Museum für Naturkunde, Berlin, Germany) and one anonymous reviewer who provided useful comments that improved the manuscript.
References
Arillo, A. 2007. Paleoethology: fossilized behaviours in amber. Geologica Acta 5: 159–166.
Arroyo-Sánchez, D.K., Víctor, J., Córdova-Tabaresa, V.M., Hernández-Damián, A.L., and Estrada-Ruiz, E. 2023. Entomopathogenic fungi (Ascomycota: Hypocreales) on a termite (Blattodea: Mastotermitidae) trapped in Miocene amber from Chiapas, Mexico. Historical Biology [published online, https://doi.org/10.1080/08912963.2023.2291769]. Crossref
Barden, P., Vincent, P., and Wang, B. 2020. Specialized predation drives aberrant morphological integration and diversity in the earliest ants. Currently Biology 30: 3818–3824. Crossref
Belokobylskij, S.A., Dubovikoff, D.A., Manukyan, A.R., and Zharkov, D.M. 2021. Braconid parasitoids of ants (Hymenoptera, Braconidae, Euphorine, Neoneurini) from Baltic amber with a discussion of records of fossil larvae parasitizing ant workers. Journal of Hymenoptera Research 84: 29–43. Crossref
Benavides, L.R. and Hormiga, G. 2020. A morphological and combined phylogenetic analysis of pirate spiders (Araneae, Mimetidae): evolutionary relationships, taxonomy, and character evolution. Invertebrate Systematics 34: 144–191. Crossref
Benjamin, S.P. and Zschokke, S. 2003. Webs of theridiid spiders: construction, structure and evolution. Biological Journal of the Linnean Society 78: 293–305. Crossref
Blackledge, T.A., Summers, A.P., and Hayashi, C.Y. 2005. Gumfooted lines in black widow cobwebs and the mechanical properties of spider capture silk. Zoology 108: 41–46. Crossref
Carbot-Chanona, G., Rivera-Velázquez, G., Jiménez-Hidalgo, E., and Reynoso, V.H. 2020. The first Pan-Carettochelys turtle in the Neogene of the American continent and its paleobiogeographical relevance. Journal of South American Earth Sciences 104: 102925. Crossref
Cevallos-Ferriz, S.R. and Ramírez, J.L. 1998. Las plantas con flores en el registro fósil. Ciencias 52: 46–57.
Cordova-Tabares, V.M., Riquelme, F., Varela-Hernández, F., and Estrada-Ruiz, E. 2024. Spider-ant predation in Oligo-Miocene Mexican amber. Palaeoentomology 7: 435–442. Crossref
Coty, D., Aria, C., Garrouste, R., Wils, P., Legendre, F., and Nel, A. 2014. Ther first ant-termite syninclusion in amber with CT-Scan analysis of taphonomy. PLoS ONE 9 (8): e104410. Crossref
Czajka, M. 1963. Unknown facts of the biology of the spider Ero furcata (Villers) (Mimetidae, Araneae). Polskie Pismo Entomologiczne 33: 229–231.
de la Fuente, J., Estrada-Peña, A., Labruña, M.B., and Szabó, M.P.J. 2024. Interaction between spiders and ticks—ancient arthropod predatory behavior? Parasitology Research 123: art. 264. Crossref
Dood, J.R. and Stanton, R.J. 1981. Paleoecology, Concepts and Applications. 502 pp. John Wiley and Sons, Chichester.
Dunlop, J.A., Selden, P.A., Pfeffer, T., and Chitimia-Dobler, L. 2018. A Burmese amber tick wrapped in spider silk. Cretaceous Research 90: 136–141. Crossref
Eberhard, W.G., Agnarsson, I., and Levi, H.W. 2008. Web forms and phylogeny of theridiid spider (Araneae: Theridiidae): chaos from order. Systematics and Biodiversity 6: 415–475.Crossref
Foelix, R.F. 1982. Biology of Spiders. 432 pp. Harvard University Press, Cambridge.
Frost, S.H. and Langenheim, R.L. 1974. Cenozoic Reef Biofacies, Tertiary Larger Foraminifera and Scleractinian Corals from Chiapas, Mexico. 388 pp. Northern Illinois University Press, De Kalb, Illinois.
García-Villafuerte, M.A. 2018a. Un posible caso de depredación preservado en ámbar de Chiapas, México: una araña (Araneae, Theridiidae) y su presa, una mosca (Diptera, Dolichopodidae). Revista Ibérica de Aracnología 33: 55–62.
García-Villafuerte, M.Á. 2018b. Primer registro fósil de un lapsino (Araneae, Salticidae) en el ámbar de Chiapas, México. Boletín de la Sociedad Geológica Mexicana 70: 689–708. Crossref
García-Villafuerte, M.A. 2022. Nuevos registros de arañas fósiles en ámbar de Chiapas, México: Thymoites carboti sp. nov. (Araneae, Theridiidae) y Mimetus sp. (Araneae, Mimetidae). Boletín de la Sociedad Geologica Mexicana 74 (2): A040622. Crossref
González-Guarda, E.A. 2019. La autoecología de los gonfoterios (Gomphotheriidae) del Pleistoceno superior de Chile. 320 pp. Doctoral Thesis, Universidad Complutense de Madrid, Facultad de Ciencias Geológicas. Madrid.
Guarisco, H. 2000. Three cobweb spider genera (Anelosimus, Tidarren, and Thymoites) and Argyrodes fictilium (Araneae:Theridiidae) recently discovered in Kansas. Journal of the Kansas Entomological Society 73 (3): 155–163.
Haug, J.T., Kiesmüller, C., Haug, G.T., Haug, C., and Hörnig, M.K. 2023. A fossil aphidlion preserved together with its prey in 40 million-year old Baltic amber. Palaeobiodiversity and Palaeoenvironments 103: 155–163. Crossref
Jackson, R.R. 1992. Eight-legged tricksters. BioScience 42: 590–598. Crossref
Jackson, R.R. and Hallas, S.E.A. 1986. Comparative biology of Portia africana, P. albimana, P. fimbriata, P. labiata, and P. shultzi, araneophagic, web-building jumping spiders (Araneae: Salticidae): Utilisation of webs, predatory versatility, and intraspecific interactions. New Zealand Journal of Zoology 13: 423–489. Crossref
Jackson, R.R. and Whitehouse, M.E.A. 1986. The biology of New Zealand and Queensland pirate spiders (Araneae, Mimetidae): aggressive mimicry, araneophagy, and prey-specialisation. Journal of Zoology 201: 279–303. Crossref
Kloock, C.T. 2001. Diet and insectivory in the ‘‘araneophagic’’ spider, Mimetus notius (Araneae: Mimetidae). American Midland Naturalist 146: 424–428. Crossref
Kloock, C.T. 2012. Natural history of the pirate spider Mimetus hesperus (Araneae; Mimetidae) in Kern Country, California. The Southwestern Naturalist 57: 417–420. Crossref
Koteja, J. 1996. Syninclusions. Inclusion-Wrostek 22: 10–12. Crossref
Langenheim, J.H., Hackner, B.L., and Bartlett, A. 1966. Mangrove pollen at the depositional site of Oligo-Miocene amber from Chiapas, Mexico. Botanical Museum Leaflets 21 (10): 289–324. Crossref
Martínez-Delclós, X., Briggs, D.E.G., and Peñalver, E. 2004. Taphonomy of insects in carbonates and amber. Palaeogeography, Palaeoclimatology, Palaeoecology 203: 19–64. Crossref
Pekár, S., Šoltysová, V., Booysen, R., and Arnedo, M. 2024. Evolution of spider- and ant-eating habits in crab spiders (Araneae: Thomisidae). Zoological Journal of the Linnean Society [published online, https://doi.org/10.1093/zoolinnean/zlae068] Crossref
Penney, D. 2014. Predatory behaviour of Cretaceous social orb-weaving spiders: comment. Historical Biology 26: 132–134. Crossref
Penney, D. and Selden, P.A. 2011. Fossil Spiders: The Evolutionary History of a Mega-diverse Order. 128 pp. Siri Scientific Press, Manchester.
Penney, D., McNeil, A., Green, D.I., Bradley, R., Withers, P.J., and Preziosi, R.F. 2012. The oldest fossil pirate spider (Araneae: Mimetidae), in uppermost Eocene Indian amber, imaged using X-ray computed tomography. Bulletin British Arachnology Society 15: 299–302. Crossref
Plotnick, R.E. and Baumiller, T.K. 2000. Invention by evolution: functional analysis in paleobiology. In: D.H. Erwin and S.L. Wing (eds.), Deep Time. Paleobiology’s perspective. Supplement to Paleobiology 26: 305–321. Crossref
Poinar Jr., G., and Buckley, R. 2014. Predatory behaviour of Cretaceous social orb-weaving spiders: response to Penney. Historical Biology 26: 135–136. Crossref
Poinar Jr., G. and Buckley, R. 2012. Predatory behaviour of the social orb-weaver spider, Geratonephila burmanica n. gen., n. sp. (Araneae: Nephilidae) with its wasp prey, Cascoscelio incassus n. gen., n. sp. (Hymenoptera: Platygastridae) in Early Cretaceous Burmese amber. Historical Biology 24: 519–525. Crossref
Poinar Jr., G. and Miller, J.C. 2002. First fossil record of endoparasitism of adult ants (Formicidae: Hymenoptera) by Braconidae (Hymenoptera). Annales of the Entomological Society of America 95: 41–43. Crossref
Romero, C. and Flórez, E. 2014. Un caso de araneofagia de Mimetus sp. (Araneae, Mimetidae), sobre Leucauge sp. (Araneae, Tetragnathidae) en el nororiente de Colombia. Revista Ibérica de Aracnología 24: 102–103.
Santos-González, A. 2005. Arañas tejedoras asociadas a los manglares de la costa de Chiapas. 137 pp. Universidad Nacional Autónoma de México, Facultad de Ciencias, México D.F.
Solórzano-Kraemer, M.M. and Brown, B. 2017. Dohrniphora (Diptera: Phoridae) from the Miocene Mexican and Dominican ambers with a paleobiological reconstruction. Insect, Systematic and Evolution 49: 299–327. Crossref
Vega, F.J., Nyborg, T., Coutiño, M.A., Solé, J., and Hernández-Monzón, O. 2009. Neogene Crustacea from southeastern Mexico. Bulletin of the Mizunami Fossil Museum 35: 51–69.
Vizcaíno, S.F., Bargo, M.S., and Cassini, G.H. 2006. Dental occlusal surface area in relation to food habits and other biologic features in fossil Xenarthrans. Ameghiniana 43: 11–26.
Webb, S.D., Beatty, B.L., and Poinar Jr., G. 2003. New evidence of Miocene Protoceratidae including a new species from Chiapas, Mexico. Bulletin of the American Museum of Natural History 279: 348–367. Crossref
Weitschat, W. and Wichard, W. 1998. Atlas der Pflanzen und Tiere im Baltischen Bernstein. 256 pp. Verlag Dr. Friedrich Pfeil, München.
Wunderlich, J. 2004. Fossil spiders in amber and copal. Beiträge zur Araneologie 5: 1–870.
Wunderlich, J. 2008. Fossil and extant and fossil (Eocene) European comb-footed spiders (Araneae: Theridiidae), with notes on their subfamilies, and with descriptions of new taxa. Beiträge zur Araneologie 5: 140–469.
Zschokke, S. 2003. Spider-web silk from the Early Cretaceous. Nature 424: 636–637. Crossref
Zschokke, S. 2004. Glue droplets in fossil spiders webs. In: D.V. Logunov and D. Penney (eds.), European Arachnology 2003 (Proceedings of the 21st European Colloquium of Arachnology, St.-Petersburg, 4–9 August 2003). Arthropoda Selecta, Special Issue 1: 367–374.
Acta Palaeontol. Pol. 69 (4): 677–685, 2024
https://doi.org/10.4202/app.01193.2024