

Biting midges from early Eocene amber of Oise, France, have no connection to other Eocene faunas
RYSZARD SZADZIEWSKI, MAXIME SANTER, ANDRE NEL, EWA KRZEMIŃSKA, KATARZYNA KOPEĆ, WIESŁAW KRZEMIŃSKI, and AGNIESZKA SOSZYŃSKA
Szadziewski, R., Santer, M., Nel, A., Krzemińska, E., Kopeć, K., Krzemiński, W., and Soszyńska, A. 2026. Biting midges from early Eocene amber of Oise, France, have no connection to other Eocene faunas. Acta Palaeontologica Polonica 71 (1): 51–68.
This study presents a study of biting midge (Diptera: Ceratopogonidae) fauna from early Eocene amber of Oise, Paris Basin, France. The fauna comprise eight species, all of them new to science: Eosphaeromias eocenicus Szadziewski, Santer, Nel, & Krzemińska gen. et sp. nov. Leptoconops oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., Brachypogon (Isohelea) europaeus Szadziewski, Santer, Nel, & Krzemiński sp. nov.; Brachypogon (Isohelea) oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov.; Brachypogon (Brachypogon) parisiensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., Stilobezzia (Acanthohelea) eocenica Szadziewski, Santer, Nel, & Krzemiński sp. nov., Forcipomyia oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., and Dasyhelea eocenica Szadziewski, Santer, Nel, & Krzemiński sp. nov. The fauna of biting midges from early Eocene Oise forests (53 Ma) shows a low diversity and has no common species with the well-studied middle Eocene fauna of Baltic amber forests (c. 40 Ma).
Key words: Diptera, Ceratopogonidae, new genus, new species, palaeoecology, Ypresian.
Ryszard Szadziewski [ryszard.szadziewski@ug.gda.pl; ORCID: https://orcid.org/0000-0002-2899-6861 ], Department of Invertebrate Zoology and Parasitology, University of Gdańsk, Wita Stwosza 59, 80-308 Gdańsk, Poland.
Maxime Santer [msantesa13@alumnes.ub.edu; ORCID: https://orcid.org/0000-0002-3685-4716 ], Departament de Dinàmica de la Terra i de l’Oceà, Facultat de Ciѐncies de la Terra, Universitat de Barcelona, Martí i Franqués, s/n, 08028 Barcelona, Spain.
Andre Nel [anel@mnhn.fr; ORCID: https://orcid.org/0000-0002-4241-7651 ], Institut de Systématique, Évolution, Biodiversité (ISYEB) Muséum national d’Histoire naturelle, CNRS, Sorbonne Université, EPHE, Université des Antilles, CP50, 57 rue Cuvier 75005, Paris, France.
Ewa Krzemińska [ekrzeminska9@gmail.com; ORCID: https://orcid.org/0000-0002-3431-9963 ] (corresponding author), Katarzyna Kopeć [kopeck1981@gmail.com; ORCID: https://orcid.org/0000-0001-6449-3412 ], and Wiesław Krzemiński [wieslawk4@gmail.com; ORCID: https://orcid.org/0000-0001-5685-891X ], Institute of Systematics and Evolution of Animals, Polish Academy of Sciences, Sławkowska 17, 31-016 Kraków, Poland.
Agnieszka Soszyńska [agnieszka.soszynska@biol.uni.lodz.pl; ORCID: https://orcid.org/0000-0002-2661-6685 ], Department of Invertebrate Zoology and Hydrobiology, University of Lodz, Banacha12/16, 90-237 Łódź, Poland.
Received 13 November 2024, accepted 22 October 2025, published online 3 March 2026.
Copyright © 2026 R. Szadziewski et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
The Ceratopogonidae is a well-known family of nematocerous flies reported from rock deposits as compression/impression fossils and from ambers as inclusions. In rock deposits they are barely preserved and details of their morphology are usually not visible. The oldest biting midge was described by Borkent et al. (2013) from a single wing preserved in the sediments of the Lower Cretaceous Purbeck Limestone Group (Dorset, UK, 142 Ma; Ogg et al. 2008). Ceratopogonids preserved with bodies are present as inclusions in no less than 15 fossil resins from the Cretaceous and Cenozoic of Europe, Asia, Central and North America (for review see Szadziewski 2018). Their record in amber covers c. 100 million years, i.e., from Lebanese amber (129–125 Ma; Maksoud and Azar 2020) to Dominican amber (20–15 Ma; Iturralde-Vinent and Mc Phee 1996). Due to excellently preserved details in resins, the morphology of these small flies was described based on multiple characters of the head, wing, thorax, legs and genitalia. This enabled reliable comparison with Recent genera (Szadziewski 1988, 1996; Szadziewski and Grogan 1998a, b) and indication of the taxa characteristic of geological ages of amber and deposits (Szadziewski 2018).
Among the ceratopogonid inclusions, the Oise amber (France) was until now not represented. This amber is of particular importance among the European Eocene resins, because its age is well established (unlike that of Baltic amber, an immense source of the dipteran inclusions). Commonly known as Oise amber, the amber from this outcrop is found in a layer of thick brown sand (Nel and Brasero 2010). The palaeoenvironment has been inferred as a forest in a fluvio-lacustrine setting formed by a fluvial system with multiple channels and ponds, without apparent marine influence, and under a warm and wet seasonal climate (Nel et al. 2004). The diverse record of fossil vertebrates (including bones, teeth, and coprolites) recovered from the layer has enabled us to correlate it with the lowermost Eocene reference locality (MP7) of Dormaal (Belgium) (Nel et al. 1999, 2004). The outcrop is dated as Sparnacian (Ypresian) of the earliest Eocene age, c. 53 Ma (Nel et al. 1999, 2004; Nel and Brasero 2010). The Oise amber is usually yellowish and translucent, and it has yielded an important collection of more than 20,000 bioinclusions, including at least 17 insect orders, mites, spiders, pseudoscorpions, mammalian hairs, and bird feathers (Nel et al. 2004; Brasero et al. 2009). The wood structure of the resin-producing tree is very similar to that of the Recent genus Daniellia within the tribe Detarieae (Fabaceae: Caesalpinioideae), based on amber pieces associated with wood fragments (de Franceschi and de Ploëg 2003; Nel and Brasero 2010). This result is supported by geochemical analyses of the amber, which shows affinities with the Caesalpinioideae within the Fabaceae (Nohra et al. 2015). The outcrop does not present the characteristics of an autochthonous amber site (Álvarez-Parra et al. 2021), as amber, plant, and vertebrate macro-remains show evidence of at least local transport, while carbonised wood could have undergone lengthy transport (Nel et al. 2004). Therefore, the Oise amber deposit might be composed of parautochthonous and allochthonous fragments.
In summary, the fauna and flora of the amber from Le Quesnoy at the Oise department, Paris Basin, are relatively well documented. Amber from Le Quesnoy, usually addressed as Oise amber, is of earliest Eocene age (Ypresian or Sparnacian, 53 Ma). An inventory list of taxa described from Oise amber was given by Brasero et al. (2009).
Institutional abbreviations.—MNHN, Muséum National d’Histore Naturelle, Paris, France.
Other abbreviations.—AR, ratio of the combined length of flagellomeres 9–13 to combined length of flagellomeres 1–8 (in female); CR, length ratio of costa to wing; TR(1/2/3), length ratio of tarsomere 1 to tarsomere 2 of foreleg (1), midleg (2) and hind leg (3). CuA, M, R, cubitoanal, medial, radial wing vein, respectively.
Nomenclatural acts.—This published work and the nomenclatural acts it contains have been registered in Zoobank: urn:lsid:zoobank.org:pub:5D56C27E-10CC-443F-B95F A3B989E44E78.
Material and methods
In total, 83 specimens of biting midges were examined. Observations and documentation were made using Olympus optical microscopes. The photographs were taken using Leica DM6000 attached to Leica M205A microscope under control the LAS Montage multifocus. The generic assignment was based on diagnostic characters used for determination of extant biting midges. For general morphological terminology of the family and keys to genera for adults see Szadziewski (1988), Szadziewski et al. (1997), and Borkent (2017).
Measurements.—Wing length is measured from basal arculus to the wing tip. Length of tarsomere 2 in fossil biting midges is measured excluding basal portion hidden in tarsomere 1. For other measurements see the Abbreviations above.
Systematic palaeontology
Order Diptera Linnaeus, 1758
Family Ceratopogonidae Newman, 1834
Subfamily Leptoconopinae Noé, 1907
Genus Leptoconops Skuse, 1889
Type species: Leptoconops stygius Skuse, 1889, New South Wales, Australia, Recent.
Leptoconops oisensis
Szadziewski, Santer, Nel,
& Krzemiński sp. nov.
Fig. 1.
Zoobank LSID: urn:lsid:zoobank.org:act:06E36370-3131-4F76-9F1D-F250D31E42F9.
Etymology: In reference to the source of this species, Oise amber.
Type material: Holotype female MNHN.F.A95076 (PA 3298). Paratype male MNHN.F.A95077 (PA 16618) (syninclusion: one specimen of Thysanoptera).
Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The female of a new species differs from other Eocene congeners in having simple claws and spherical flagellomeres 7–11. Male has unique tergite 9 with two slender apicosubmedian processes.
Description.—Male complete, dark, embedded in dark amber, hence poorly visible (Fig. 1A1). Antenna with 13 flagellomeres, last one elongated; total length about 0.39 mm, plume well developed. Proboscis very short. Palpus with palpomeres 4 and 5 fused. Palpomere 3 barely visible, without sensory pit. Wing length 0.61 mm, costa very short, membrane without macrotrichia. Legs slender, without modifications. TR(2) 1.9, TR(3) 1.6. Genitalia (Fig. 1B2). Gonocoxite with enlarged base. Gonostylus almost straight, armed with distinct apical tooth. Tergite 9 with two slender, pointed, apicosubmedian processes.

Fig. 1. Ceratopogonid biting midge Leptoconops oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov. (Oise amber, Le Quesnoy, France; lower Eocene). A. MNHN.F.A95077 (PA 16618), paratype male; specimen in lateral view (A1), genitalia in ventral view (A2). B. MNHN.F.A95076 (PA 3298), holotype female; specimen in lateral view (B1), drawings of flagellum (B2), distal palpomeres (B3), and cerci (B4) in lateral views. Abbreviations: p3, palpomere 3; p4+5, fused palpomeres 4+5.
Female complete, dark, barely visible in dark amber (Fig. 1B1). Body length without flagellum 1.2 mm. Flagellum length 0.28 mm, composed of 12 flagellomeres of which 2–11 are more or less spherical, and flagellomere 12 about 3 times longer than preceding one (Fig. 1B2). Proboscis short, mandible with numerous small teeth. Palpal segment 3 about 0.055 mm long, with indistinct depression on ventral surface covered with sensilla capitata, primitive palpomeres 4 and 5 fused (Fig. 1B3). Wing length 0.60 mm long, with short costal vein, membrane without macrotrichia. Legs slender, TR(1) 1.9, TR(3) 2.1. Claws simple, without teeth. Cerci relatively short (Fig. 1B4).
Remarks.—The subgeneric position of the new species is not clear. Female of this species has 12 flagellomeres like members of the subgenus Leptoconops Skuse, 1889, or Proleptoconops Clastrier, 1974; however, the male has tergite 9 with apicolateral processes in a submedian position like males of the subgenus Holoconops Kieffer, 1918. Species of Leptoconops are reported from middle Eocene Baltic amber from Bay of Gdańsk, Poland (Leptoconops succineus Szadziewski, 1988) and from Rovno, Ukraine (Leptoconops rovnensis Sontag & Szadziewski, 2011). Leptoconops rovnensis differs in having female claws with distinct basal tooth and flagellomere 11 slightly cylindrical. The female of L. succineus has simple claws like in the new species, however differs in having flagellomeres 7–11 cylindrical which in L. oisensis are spherical.
Species of the genus Leptoconops are good indicators of sea shore or estuarine environments near amber forests, as their larvae usually live in sand of coastal and inland beaches, indicating that coastal or estuarine ecosystems close to amber forests were present. Leptoconops is a relictual pantropical genus that was distributed worldwide during the Cretaceous (Szadziewski 2018).
Stratigraphic and geographic range.—Type locality and horizon only.
Subfamily Ceratopogoninae Newman, 1834
Tribe Ceratopogonini Newman, 1834
Genus Brachypogon Kieffer, 1899
Type species: Ceratopogon vitiosus Winnertz, 1852, Germany, Recent.
Subgenus Isohelea Kieffer, 1917b
Type species: Psilohelea sociabilis Goetghebuer, 1920, Belgium, Recent.
Remarks.—Our material constitutes the oldest record of the genus and subgenus. Previously the genus was recorded from middle Eocene and Miocene (Szadziewski 2018).
Brachypogon (Isohelea) europaeus Szadziewski, Santer, Nel, & Krzemiński sp. nov.
Fig. 2.
Etymology: In reference to Europe, the continent of its occurrrence.
Zoobank LSID: urn:lsid:zoobank.org:act:395D4464-5B68-4A06-A2 EE-BDD7296CF632.
Type material: Holotype male, MNHN.F.A95078 (PA8129). Paratype male, MNHN.F.A95079 (PA12805).
Type locality: Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The only species of Ceratopogonidae having male flagellum composed of 13 separated flagellomeres, both first radial cells, and male genitalia stout with very long apicolateral processes. Female unknown.
Description.—Male complete (Fig. 2A1, B1). Flagellum composed of 13 separated flagellomeres, distal 3 elongated, total length 0.34 mm. Proboscis short. Palpus five-segmented. Palpomere 3 rather stout, probably with sensory pit. Scutellum with four marginal setae. Wing length 0.60–0.67 mm, CR 0.47–0.49; two small radial cells present, wing membrane without macrotrichia. Legs slender. Tarsomere 1 of hind leg with unmodified row of palisade setae. TR(3) 2.0. Genitalia stout (Fig. 2A, B1). Sternite 9 barely visible, rather with shallow caudomedian excavation. Tergite 9 short with very long cylindrical apicolateral processes. Gonocoxite stout. Gonostylus stout, shorter than gonocoxite. Aedeagus short, triangular with low basal arch. Parameres present, barely visible.
Female unknown.

Fig. 2. Ceratopogonid male biting midge Brachypogon
(Isohelea) europaeus
Szadziewski, Santer, Nel, & Krzemiński sp. nov. (Oise amber, Le
Quesnoy, France; lower Eocene). A.
MNHN.F.A95078 (PA12805), paratype; body in lateral view (A1),
genitalia in dorsal view (A1).
B. MNHN.F.A95078
(PA 8129), holotype, body in lateral view (B1);
genitalia in ventral view (B2),
gonostyles and tergite 9 not shown.
Remarks.—Brachypogon eocenicus Szadziewski, 1988, is known from the Baltic amber, with similar genitalia armed with very long apicolateral processes and with separated flagellomeres. However, in B. eocenicus only the first radial cell is developed while in the new species both first radial cells are present.
Stratigraphic and geographic range.—Type locality and horizon only.
Brachypogon (Isohelea) oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov.
Fig. 3.
Zoobank LSID: urn:lsid:zoobank.org:act:93159D14-B6B7-4AA7-92 AB-C0DBE30E6F3F.
Etymology: In reference to the source of this species, Oise amber.
Type material: Holotype male, MNHN.F.A95080 (PA12572).
Type locality: Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The male of the species is unique among Ceratopogonidae in having antenna with 10 flagellomeres and gonostyli with slender cylindrical distal half. Female unknown.
Description.—Male well preserved, complete, dark (Fig. 3A1). Body length 1.1 mm. Eyes separated. Flagellum 0.37 mm long; with 10 flagellomeres, proximal 1–7 short (0.12 mm), distal 3 elongated (0.15 mm). Palpus five-segmented, palpomere 3 swollen, sensorium not visible, combined length of palpomeres 3–5 about 0.075 mm. Scutellum with four or more marginal bristles. Wing length 0.51 mm, CR 0.49. Both first radial cells present, first one slit-like, second one short, almost rectangular (Fig. 3A3). Wing membrane with few macrotrichia in 1 row along apical margin (Fig. 3A2). Legs slender, unarmed. Palisade setae on tarsomere 1 of hind leg present, barely visible. Hind tibial comb with at least seven spines. Tarsomeres 4 slightly subcylindrical. TR(1) 2.5, TR(2) 2.4, TR(3) 2.4.
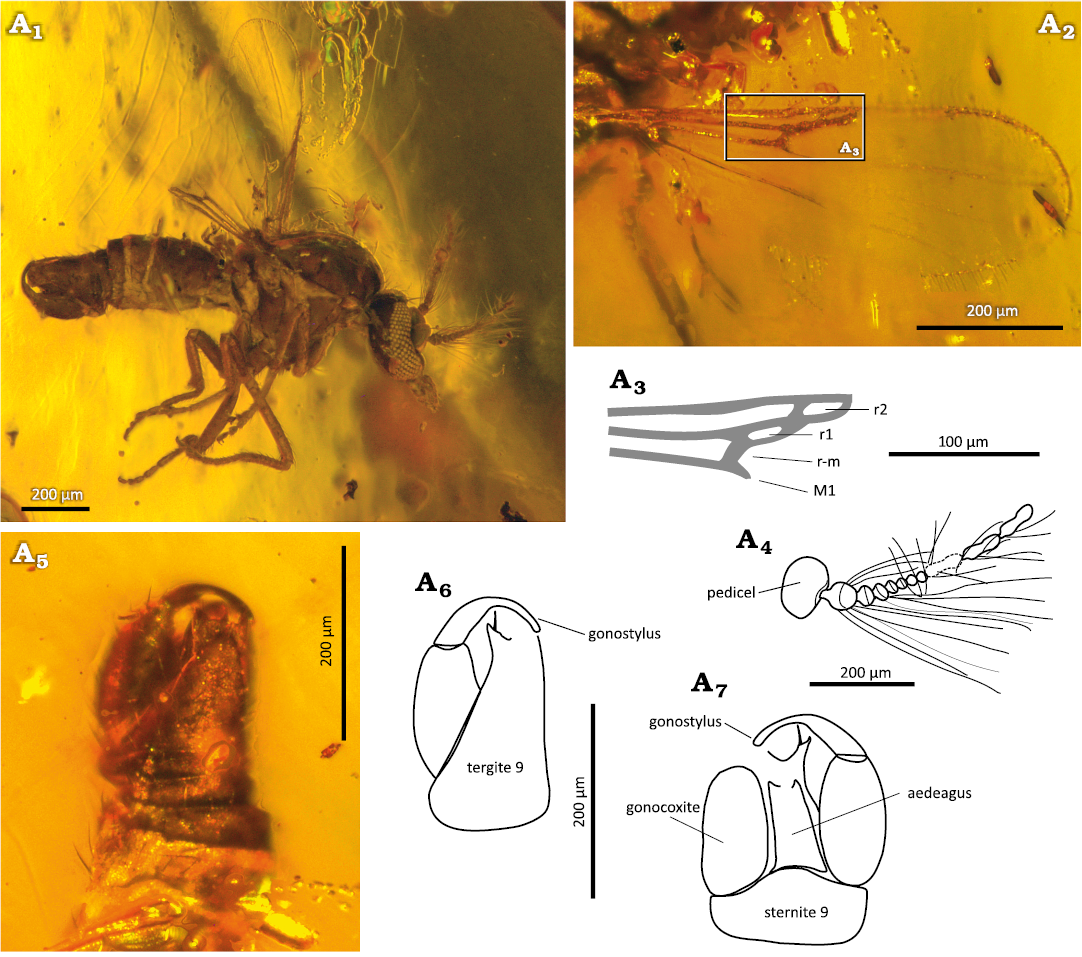
Fig. 3. Ceratopogonid male biting midge Brachypogon (Isohelea) oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., holotype MNHN.F.A95080 (PA 12572) from Oise amber, Le Quesnoy, France; lower Eocene. A1, habitus in lateral view; A2, wing in ventral view; A3, first radial cells (reconstruction of fragment marked A2); A4, antenna in lateral view; genitalia: photo (A5) and reconstruction (A6) laterodorsally, reconstruction ventrally (A7). Abbreviations: r-m, radial-medial crossvein; M1, medial vein; r1, r2, radial cells.
Genitalia large, slightly rotated (Fig. 3A1, A4–A6). Sternite 9 with caudomedian margin slightly convex. Tergite 9 long, with long subcylidrical apicolateral processes armed with pointed apical projection and 1 strong seta. Gonocoxite subcylindrical, about 2 times longer than broad, unmodified. Gonostylus C-curved, long; proximal half stout, distal half slender and cylindrical (Fig. 3A4–A6). Aedeagus plate-like, long and narrow (Fig. 3A6). Parameres not visible.
Female unknown.
Stratigraphic and geographic range.—Type locality and horizon only.
Subgenus Brachypogon Kieffer, 1899
Type species: Ceratopogon vitiosus Winnertz, 1852, Germany, Recent.
Brachypogon (Brachypogon) parisiensis Szadziewski, Santer, Nel, & Krzemiński sp. nov.
Fig. 4.
Zoobank LSID: urn:lsid:zoobank.org:act:E083EE79-D21C-4D52-9764- A3071D869834.
Etymology: In reference to the Paris Basin in France where Oise amber comes from.
Type material: Holotype male, MNHN.F.A95081 (PA-Di-1024).
Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The only species of Ceratopogonidae with both first radial cells fused, flagellum composed of 13 flagellomeres, 2–11 fused, very short palpomere 3, and slender gonostylus.
Description.—Male barely visible (Fig. 4). Proboscis short. Palpomere 3 very short, as long as wide, about 0.022 mm long. Antenna with 13 flagellomeres, total length 0.30 mm; flagellomeres 2–11 fused. Wing length 0.50 mm, CR 0.50, both first radial cells fused, wing membrane without macrotrichia. Genitalia thin, barely visible. Tergite 9 with distinct cylindrical apicolateral processes. Gonostylus slender, C-curved.
Female unknown.

Fig. 4. Ceratopogonid male biting midge Brachypogon (Brachypogon) parisiensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., holotype MNHN.F.A95081 (PA-Di-1024) in dorsal view; Oise amber, Le Quesnoy, France; lower Eocene. Abbreviations: r-m, radial-medial crossvein; R1-3, radial vein.
Remarks.—Two species of the subgenus with totally reduced both first radial cells are reported from Baltic amber: Brachypogon balticus Szadziewski, 1988, and B. gedanicus Szadziewski, 1988 (Szadziewski 1988). Male genitalia of the new species resemble those of B. gedanicus, however both species differ in number of male flagellomeres: 10 in B. gedanicus, 13 in B. parisiensis, sp. nov.
Stratigraphic and geographic range.— Type locality and horizon only.
Genus Stilobezzia Kieffer, 1911a
Type species: Stilobezzia festiva Kieffer, 1911a, India, Recent.
Subgenus Acanthohelea Kieffer, 1917a
Type species: Acanthohelea pruinosa Kieffer, 1917a, New South Wales, Australia, Recent.
Stilobezzia eocenica Szadziewski, Santer, Nel, & Krzemiński sp. nov.
Fig. 5.
Zoobank LSID: urn:lsid:zoobank.org:act:FAD607C7-B9D2-4F12-BD 65-8BCD09DB34A7.
Etymology: In reference to the Eocene epoch.
Type material: Holotype MNHN.F.A95082 (PA 1898), female, in two pieces of amber with well visible wings and legs. Paratype MNHN.F.A95083 (PA 1898), female, complete in one piece of amber.
Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The only fossil species of the genus with macrotrichia on wing membrane and the female legs with a pair of long spines (batonnets) on tarsomere 5. Male unknown.
Description.—Male unknown. Female well preserved (Fig. 5A1). Flagellum slender, length 1.05 mm, AR 1.06, composed of 13 cylindrical flagellomeres, flagellomeres 2–12 gradually increasing in length (Fig. 5A2). Proboscis long, palpus slender, five-segmented, palpomere 3 slender, sensory pit not visible. Wing length 1.32 mm, CR 0.79, macrotrichia at wing apex present, media petiolate, both first radial cells well developed (Fig. 5A3). Legs slender, femora unarmed, tarsomeres 4 cordiform, with sinuous apical seta (Fig. 5A6). Tarsomere 5 with one pair of subbasal strong slender spines, batonnets (Fig. 5A4–A6). Claws similar on all legs, single with distinct basal tooth, on outer surface, at base of talon, 2 oblique barbs present (Fig. 5A5, A6). TR(3) 2.2.
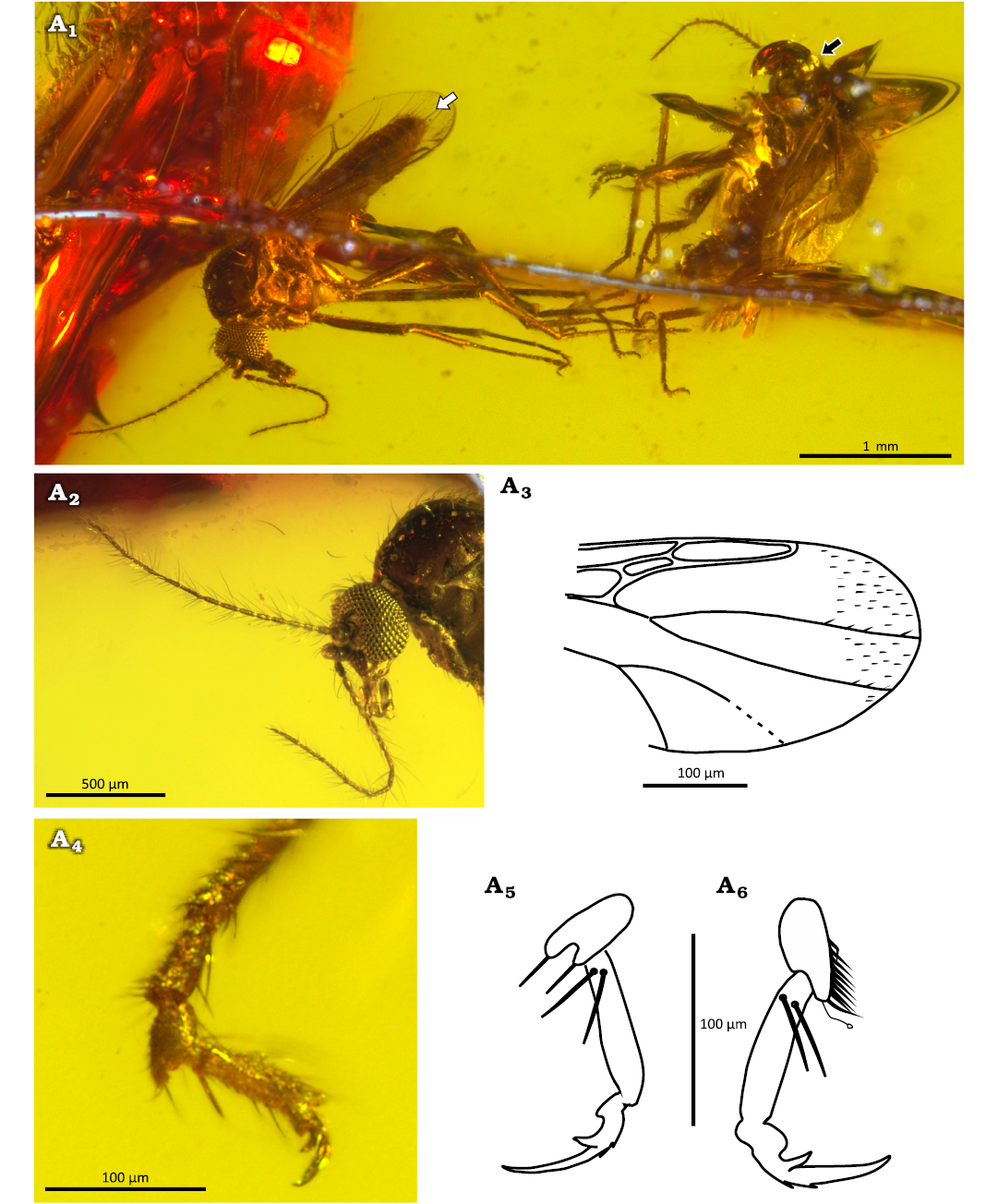
Fig. 5. Ceratopogonid female biting midge Stilobezzia (Acanthohelea) eocenica Szadziewski, Santer, Nel, & Krzemiński sp. nov. ; Oise amber, Le Quesnoy, France; lower Eocene. A1, MNHN.F.A95082 (PA 1898) holotype, habitus in lateral view (white arrow) and MNHN.F.A95083 (PA 1898) paratype, habitus in lateral view (black arrow). A2–A6, holotype MNHN.F.A95082 (PA 1898): head with antennae, laterodorsal view (A2); distal half of wing (A3); distal tarsomeres of middle leg, lateral view (A4); claw of midleg, lateral view (A5); claw of hind leg, lateral view (A6).
Remarks.—The extant genus Stilobezzia has a worldwide distribution and comprises 349 extant and 10 fossil species (Borkent and Dominiak 2020). Within the subgenus Acanthohelea tarsomeres 5 in females rarely are armed with strong ventral spines called batonnets (Das Gupta and Wirth 1968; Wirth and Grogan 1988; Szadziewski 1988). The subgenus Acanthohelea of worldwide distribution including 175 extant species is especially numerous in temperate regions (Wirth and Grogan 1988). The oldest species of the genus are known from the Late Cretaceous ambers from New Jersey (Turonian, 94–90 Ma; Kosar et al. 2024), Provence in France (Santonian, 84–86 Ma; Saint Martin et al. 2021) and Canada (79.5–76.5 Ma; Eberth and Hamblin 1993) (Szadziewski et al. 2016; Szadziewski 2018). Other species are known from the early Eocene Cambay amber (48–56 Ma; Smith et al. 2016), middle Eocene Sakhalin amber (45 Ma; Kodrul et al. 1999), middle Eocene Baltic amber (middle Eocene, 48–34 Ma; Grimaldi and Ross 2017), Miocene Dominican amber (20–15 Ma; Iturralde-Vinent and Mac Phee 1996) (Szadziewski 2018). They are also reported as rock compression fossils from the upper Eocene of Isle of Wight, England (Ross and Self 2014) and from Miocene deposits of Rott, Germany (Krzemiński et al. 2019).
Stratigraphic and geographic range.—Type locality and horizon only.
Tribe Sphaeromiini Newman, 1834 sensu lato
Remarks.—The tribe of higher predators includes many small genera of unclear phylogenetic relationships. Borkent (2014) proposed to divide the traditional tribe including species with females armed with batonnets on tarsomeres 5 into three tribes: Hebetulini Borkent, 2014, Johannsenomyiini Crampton, 1925, and Sphaeromiini Newman, 1834 sensu stricto. This proposal was based on morphology of pupae and diagnoses of proposed new tribes based on adults are weak and not well supported by adult morphology (Borkent 2014, 2017). According to the new proposal by Borkent (2014) Mallochohelea martae Szadziewski, 2005, from Eocene Baltic amber with female having the abdominal sternite 8 bearing two groups of long setae (tuft) and equal claws with external tooth is placed in tribe Johannsenomyiini.
Biting midges of the tribe Sphaeromiini sensu lato are extremely rare among fossils. This is the only second record of the tribe among amber inclusions and fossils. The new genus described below falls to the group of genera with females having tarsomeres 5 with batonnets distributed on entire length of the segment and equal claws, simple or armed with internal basal tooth is a member of the tribe Sphaeromiini sensu stricto in the limits as proposed by Borkent (2014).
The conspicuous armature of tarsi in females in this tribe is used for grasping the prey, and also for hunting the conspecific males in order to consume them instead or in course of the mating (Szadziewski and Sontag 2022). The present finding of a female, armoured with batonnets and long claws on tarsi, and spines along femora indicates that such predatory habits were present already in the earliest Eocene.
Genus Eosphaeromias Szadziewski, Santer, Nel, & Krzemińska gen. nov.
Type species: Eosphaeromias eocenicus gen. et sp. nov., monotypic; see below.
Zoobank LSID: urn:lsid:zoobank.org:act:56223E64-E881-4B00-8F10-875CA1FE 6C23.
Etymology: A combination of the Eocene epoch and the name of the extant genus Sphaeromias Curtis, 1829. Gender masculine.
Diagnosis.—As for the monotypic species.
Remarks.—The new genus has unique combination of female characters unknown among known extant genera of the group Sphaeromiini sensu lato. Most of characters listed in the diagnosis are known among other genera except for very short tarsomeres 3 and 4 on all legs and the small size of the female.
In some species of Mallochohelea Wirth, 1962, and Jenkinshelea Macfie, 1934, and some other genera tarsomeres 3 and 4 are very short on fore and mid legs, however short tarsomere 3 on hind legs is known only in Eosphaeromias Szadziewski, Santer, Nel, & Krzemińska gen. nov.
Equal claws with internal tooth (external absent) are present in seven extant genera included in the tribe Sphaeromiini by Borkent and Dominiak (2020), four other genera have simple claws. Very long internal teeth of the new genus are similar to those known in the tropical genus Mackerrasomyia Debenham, 1970, however, its females have greatly swollen fore femur armed with strong ventral spines and the abdominal sternite 8 bears long tuft setae. Equal long claws similar on all legs armed with short or long internal tooth, and pointed batonnets have females of species in the genera Austrosphaeromias Spinelli, 1997, from South America, Lanehelea Wirth & Blanton, 1972, from Colombia and Sphaeromias Curtis, 1829, of worldwide distribution. Females of Austrosphaeromias differ in having tufts of long setae on abdominal sternite 8 and swollen fore femora armed with ventral spines, while Lanehelea and Sphaeromias have smaller internal teeth and elongated tarsomeres 3 and 4, third one almost cylindrical on all legs (Wirth and Blanton 1972; Szadziewski et al. 2007). Similar large internal teeth/barbs are present in the tropical genus Neosphaeromias Das Gupta & Wirth, 1971, of the tribe Johannsenomyiini. However, females in this genus have swollen fore femur armed with numerous ventral spines and in addition their claws have external basal tooth.
Stratigraphic and geographic range.—Type locality and horizon only.
Eosphaeromias eocenicus Szadziewski, Santer, Nel, & Krzemińska sp. nov.
Figs. 6, 7.
Zoobank LSID: urn:lsid:zoobank.org:act:25A79F8B-8DCE-4AA1-B7 FA-960A0A06AD31.
Etymology: In reference to the Eocene epoch.
Type material: Holotype female, MNHN.F.A95084 (PA 185).
Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The genus and species is distinct among the tribe Sphaeromiini sensu lato in having the following combination of female characters: two claws similar on all legs, long, equal, almost straight, each with very long and stout basal internal barb; tarsomeres 4 and 3 short on all legs, legs slender, fore femur armed with some ventral spines, tarsomere 5 with 5–6 pairs of pointed batonnets distributed on entire length of the segment, abdominal sternite 8 without tuft setae; wing length 0.9 mm, both first radial cells present, CR 0.81. Male unknown.
Description.—Female complete (Fig. 6A1). Body length 1.39 mm, wing length 0.9 mm apparently making it the smallest known species within the tribe Sphaeromiini sensu lato.
Eyes separated. Flagellum composed of 13 flagellomeres, total length 0.74 mm, AR 1.46, terminal flagellomere blunt. Flagellomeres 2–8 gradually increasing in length from spherical to cylindrical, lengths of distal five flagellomeres as follows (in mm): 9, 0.074; 10, 0.088; 11, 0.081; 12, 0.092; 13, 0.097 (Fig. 6A3). Proboscis short, mouthparts unclear. Palpus five-segmented, palpomere 3 slightly swollen. Scutum without anterior spine. Scutellum with seven or more marginal bristles in one row. Paratergite broad. Wing length 0.89 mm, width c. 0.33 mm. Wing membrane covered with very small microtrichia, macrotrichia absent. Distinct sensilla spot visible between R4+5 and M1, near R4+5 distal part. Veins M1 and M2 sessile, M2 forking proximal of crossvein r-m (Fig. 6A2, 7A1); both first radial cells well developed, costal ratio about 0.81; CuA forking at the level of r-m. Legs slender, fore femur armed with two ventral spines (Fig. 7A2). Tarsomere 1 of hind leg with one row of palisade setae. Tarsomeres 3 and 4 very short on all legs, tarsomere 5 armed with 5–6 pairs of slender batonnets distributed on entire length of the segment. Claws similar on all legs, both long and equal, flattened and slightly curved, each with long and strong internal basal barb, without external basal tooth (Fig. 6A4, A5). Abdomen barely visible, long tuft setae on sternite 8 absent. Genitalia unclear, obscured by gas bubbles.
Male unknown.

Fig. 6. Ceratopogonid female biting midge Eosphaeromias eocenicus Szadziewski, Santer, Nel, & Krzemińska sp. nov., holotype MNHN.F.A95084 (PA 185); Oise amber, Le Quesnoy, France; lower Eocene. Habitus in lateral view (A1), wings in ventral view (A2), head and thorax in lateral view (A3), claws of hind legs in lateral view (A4, A5).

Fig. 7. Ceratopogonid female biting midge Eosphaeromias eocenicus Szadziewski, Santer, Nel, & Krzemińska sp. nov., holotype MNHN.F.A95084 (PA 185); Oise amber, Le Quesnoy, France; lower Eocene. Wings, upper in dorsal, lower in ventral view (A1), venation of wing fragment (A2); fore femur with visible two ventral spines (A3). Abbreviations: M1, M2, medial veins; r-m, radial-medial crossvein; r1, r2, radial cells.
Remarks.—Biting midges trapped in fossil resins generally are smaller than those preserved in rock sediments (the amber trap is biased toward small bodied insects; Solórzano Kraemer et al. 2018) and also smaller than extant species (RS, personal observation). However, Eosphaeromias eocenicus Szadziewski, Santer, Nel, & Krzemińska gen. et sp. nov. with female wing length 0.9 mm is exceptionally small and probably is the smallest known species within the tribe Sphaeromiini sensu lato The female of other fossil member of the tribe, Mallochohelea martae Szadziewski, 2005, from Baltic amber, is much bigger, with wing length 3.3 mm.
Stratigraphic and geographic range.—Type locality and horizon only.
Subfamily Forcipomyiinae Lenz, 1934
Tribe Forcipomyiini Lenz, 1934
Genus Forcipomyia Meigen, 1818
Type species: Tipula bipunctata Linnaeus, 1767, Europe, Recent.
Forcipomyia oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov.
Figs. 8, 9 .
Zoobank LSID: urn:lsid:zoobank.org:act:C0803ADD-35CD-48CC-B99 B-80C7B77CAC0E.
Etymology: In reference to the source of this species, Oise amber.
Type material: Holotype male, MNHN.F.A95085 (PA 3916). Paratypes (1 male, 5 females), all from the type locality and horizon: MNHN.F.A95086 (LQ 148) male, MNHN.F.A95087 (LQ148) female; MNHN.F.A95088 (PA 3336) female; MNHN.F.AA95089 (PA 5964) female; MNHN.F.A95090 (PA 7870), 2 females.
Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The small species having in both sexes second radial cell long, whole wing membrane covered with distinct macrotrichia, short proboscis with five-segmented palpus and high tarsal ratio of hind leg 2.3–2.8. The only species in the genus having long separated parameres in male genitalia that are curved dorsally.
Material.—64 females: MNHN.F.A95095 (LQ 97); MNHN.F.A95095 (RI 110); MNHN.F.A95096 (RI 170); MNHN.F.A95098 (PA 222) 4 specimens; MNHN.F.A95099 (PA 562) 2 specimens; MNHN.F.A95100 (PA 1391); MNHN.F.A95101 (PA 1565) 32 specimens or more in icicle; MNHN.F.A95105 (3991) 3 specimens; MNHN.F.A95106 (8547) 2 specimens; MNHN.F.A95107 (PA 15019) 9 specimens; MNHN.F.A95108 (PA 15276), with mite Acari on abdomen; MNHN.F.A95109 (PA 18801).
Description.—Male: Body slender (Fig. 8A1, A2). Flagellum composed of 13 flagellomeres, plume well developed, total length 0.79 mm, distal 4 elongated, their length in mm are as follows: 10, 0.120; 11, 0.100; 12, 0.090; 13, 0.100 (Fig. 8A3). Proboscis short, mouthparts without teeth. Palpus five-segmented. Palpomere 3 swollen on basal half, with sensory pit at mid-length, 0.085 mm long (Fig. 8A4). Wing length 0.80–1.25 mm, CR 0.66–0.68. Wing membrane covered with macrotrichia, microtrichia distinct. First radial cell small, second one long. Legs: TR(1) 2.0, TR(2) 2.1, TR(3) 2.4. Genitalia (Fig. 8A5–A8): gonocoxite slender, gonostylus slender straight (Fig. 8A5, A6). Parameres long, separated, almost hockey stick like with blunt apices directed dorsally (Fig. 8A5, A6). Aedeagus barely visible, long with forked apex, basal arch low.
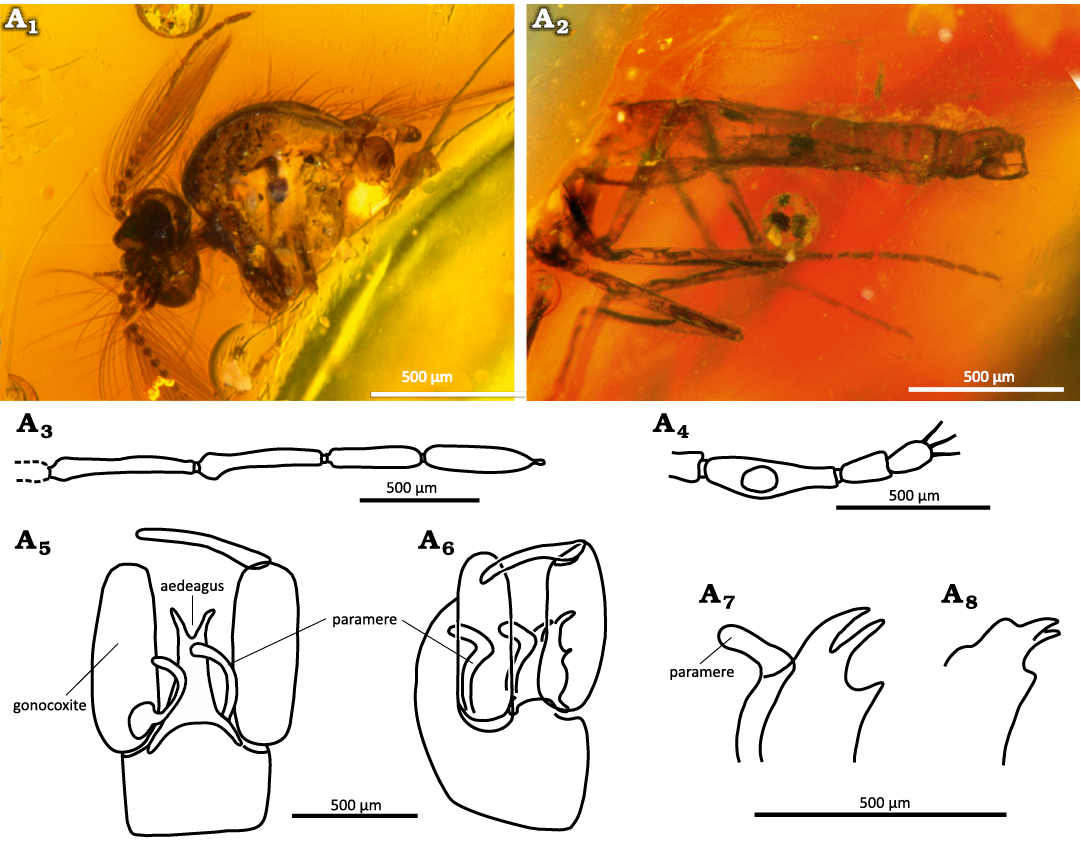
Fig. 8. Ceratopogonid male biting midge Forcipomyia oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., holotype MNHN.F.A95085 (PA3916); Oise amber, Le Quesnoy, France; lower Eocene. A1, anterior body with head in ventrolateral view; A2, posterior body with abdomen in ventrolateral view; A3, distal flagellomeres 9–13 in lateral view; A4, palpus in lateral view; genitalia, ventral (A5) and lateroventral (A6) views; A7, paramere and aedeagus, lateral view; A8, apex of aedeagus, lateral view.

Fig. 9. Ceratopogonid female biting midge Forcipomyia oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., paratype MNHN.F.A95088 (PA 3336); Oise amber, Le Quesnoy, France; lower Eocene. A1, A2, whole specimen in dorsal view; A3, wing in dorsal view; A4, first radial cells.
Female: Similar to male, with usual sexual differences (Fig. 9A1, A2). Body length 1.1–1.2 mm. Flagellum composed of 13 flagellomeres, total length 0.38 mm, AR 1.62. Proboscis very short. Palpus five-segmented, palpomere 3 stout, swollen on basal 5/6. Wing length 0.60–0.72 mm, CR 0.65–0.68. Whole wing membrane covered with macrotrichia (Fig. 9A3), both first radial cells well developed, second one about 3 times longer than first one (Fig. 9A4). TR(1) 2.8, TR(3) 2.8. Scutellum with very long and numerous marginal setae. Cerci very short.
Remarks.—Within Forcipomyia apices of parameres are straight or curved ventrally. In the new species they are curved dorsally and this shape probably is unique within the genus. The subgeneric position of the species is not clear. It is mostly similar to species of the subgenus Synthyridomyia Saunders, 1957, which have separated parameres.
Stratigraphic and geographic range.—Type locality and horizon only.
Tribe Dasyheleini Lenz, 1934
Genus Dasyhelea Kieffer, 1911b
Type species: Dasyhelea halophila Kieffer, 1911b, Croatia, Recent.
Dasyhelea eocenica Szadziewski, Santer, Nel, & Krzemiński sp. nov.
Fig. 10.
Zoobank LSID: urn:lsid:zoobank.org:act:CFF61F32-2F55-4DFB-8617- 6B3B4E76F615.
Etymology: In reference to the Eocene Epoch.
Type material: Holotype male (MNHN.F.A95091), and two male paratypes (MNHN.F.A95092 and MNHN.F.A95093) in one amber piece (4206). The holotype is positioned between two male paratypes on left and right (one paratype incomplete, without genitalia).
Type locality: Le Quesnoy, Paris Basin, France.
Type horizon: Eocene Oise amber.
Diagnosis.—The only fossil species of Ceratopogonidae with both first radial cells developed, tergite 9 with distinct apicolateral processes, and gonostylus with slender cylindrical shining black apical third. Female unknown.
Description.—Male: Body complete (Fig. 10A1). Flagellum 0.50–0.51 mm long, plume well developed, distal 4 flagellomeres elongated, flagellomere 13 with rounded apex (Fig. 10A2). Proboscis short. Palpus five-segmented; palpomere 3 short and stout (Fig. 10A3). Scutum shining black. Wing length 0.60–0.68 mm, CR 0.55–0.58. First radial cell slit-like present, second one rectangular, membrane with macrotrichia on distal half. TR(1) 2.3–2.5, TR(2) 2.2, TR(3) 2.0–2.3. Genitalia (Fig. 10A5) slightly rotated. Sternite 9 distinct, M-shaped with deep caudomedian excavation. Tergite 9 with long cylindrical apicolateral processes armed with apical seta. Gonocoxite slender, without modifications. Gonostylus almost straight stout on basal 2/3 and slender, cylindrical, shining black on apical 1/3 (Fig. 10A4). Aedeagus and parameres barely visible.
Female unknown.
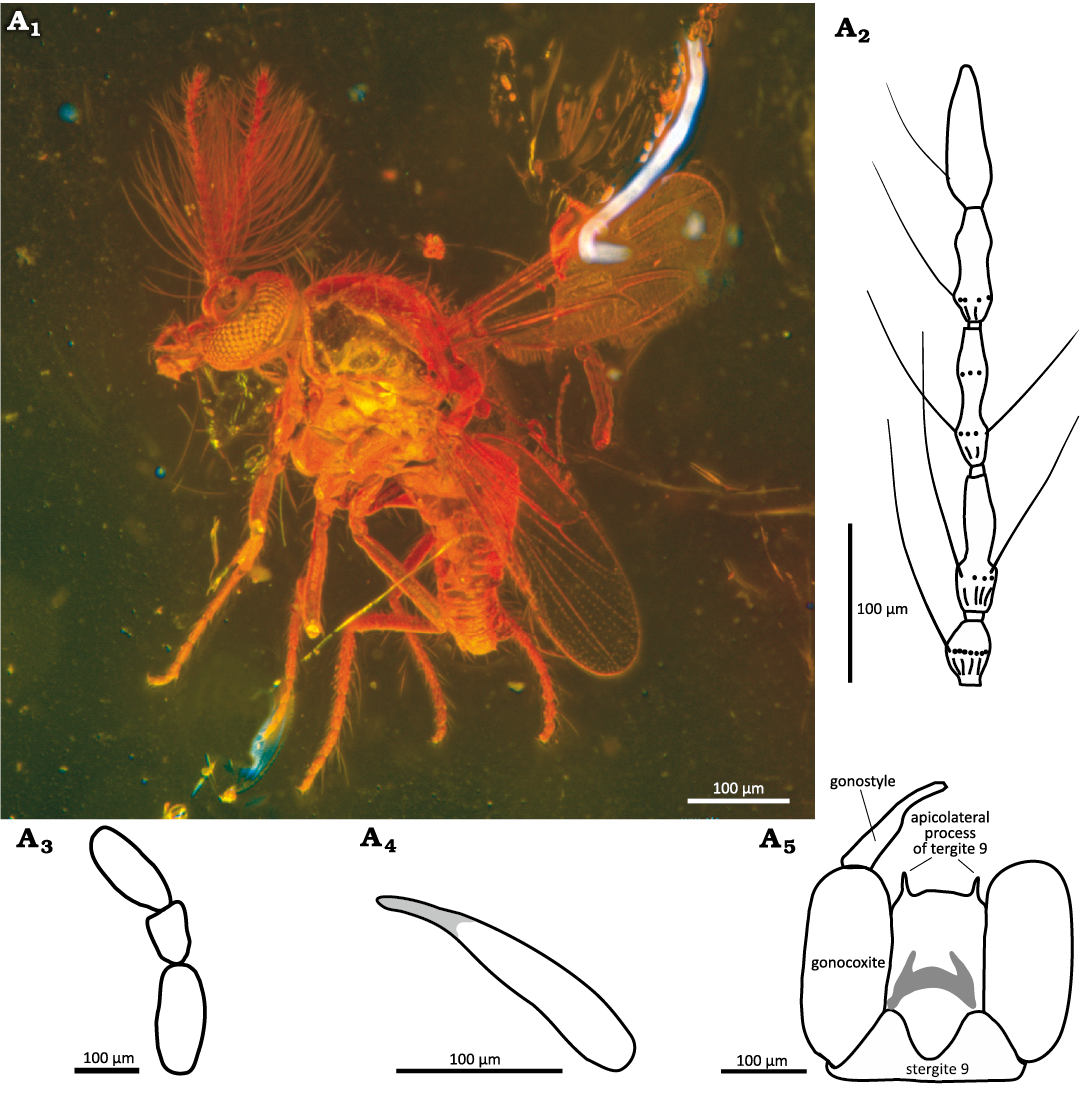
Fig. 10. Ceratopogonid male biting midge Dasyhelea oisensis Szadziewski, Santer, Nel, & Krzemiński sp. nov., holotype MNHN.F.A95091; Oise amber, Le Quesnoy, France; lower Eocene. A1, habitus in lateral to ventrolateral view; A2, distal flagellomeres 9–13 in lateral view; A3, palpomeres 3–5; A4, gonostylus; A5, genitalia in ventral view.
Remarks.—The subgeneric position of the species is not clear. The following combination of characters: two first radial cells developed, flagellomere 13 with rounded apex, abdominal tergite 9 with cylindrical apicolateral processes is unknown within typical European subgenera of the genus Dasyhelea (Dominiak and Szadziewski 2010; Dominiak 2012).
Stratigraphic and geographic name.—Type locality and horizon only.
Discussion
In 29 amber pieces of Oise amber, among 83 biting midges we found eight new species which represent three subfamilies, three tribes, and seven genera/subgenera, of which six are recently represented, and the seventh is a new extinct genus. Nevertheless, the diversity is rather low here when compared to ceratopogoniid fauna from other Eocene ambers.
The dominant genus in Oise amber is Forcipomyia, with 71 specimens (86% of all ceratopogonids; Table 1). It is less common in Baltic amber including Rovno and Bitterfeld deposits (11–32% of all biting midges; Sontag and Szadziewski 2011), but prevails in several other Cenozoic ambers (72% in Eocene Sakhalin amber: Szadziewski 1990; 41% in Eocene Indian amber: Stebner et al 2017; 67% in Miocene Mexican amber: Szadziewski and Grogan 1996; and finally 42% in Miocene Dominican amber: Szadziewski and Grogan 1998b). Noteworthy, the samples from Sakhalin amber (25 specimens), Indian amber (34) or Mexican amber (18) were very small.
Especially interesting is an icicle
containing 32 females
of Forcipomyia oisensis Szadziewski,
Santer, Nel, & Krze-miński sp. nov. Such abundance of females in a small (c. 1 cm3) piece of amber is
extremely rare and clearly refers to a specific life situation of
these flies. As a rule, the females of extant ceratopogonids (and
various nematoceran flies)
hatch some days after males and disperse in seek of swarming males.
Males usually depart from hatching places and form swarms over some
elevated structure as bush, stone or stick (a so called marker) in a
well-lit place to be easily viewed by females. Such places might be far
from resining trees. Therefore, finding of so many females trapped
together may indicate a situation just after their mass eclosion;
alternatively, a mass oviposition after their return from mating places
to the forest. As the larvae of Forcipomyia
are generally terrestial and live in rotting wood or plant debris, our
finding also shows that such habitats were common in Oise forests.
Dominance of the genus Forcipomyia in Oise amber, and especially the abundance of females, recall those observed in Dominican amber, where as many as 48 females in one amber piece were found (Szadziewski and Grogan 1998b). In both Oise and Dominican ambers this genus is dominant (86% and 41%, respectively), with females prevailing (97% and 80%, respectively) which may suggest that forests where they lived were ecologically similar. Actually, both ambers were produced by leguminous trees of the Fabaceae family, and in that they differ from Baltic amber which was produced by pine trees. In Baltic amber females constituted only 67% specimens of the genus Forcipomyia (Table 1).
Table 1. Taxonomic proportions of Eocene biting midges from amber of Oise presently examined and from Baltic amber (after Szadziewski 1988 and this paper). –, percentage omitted for too small values.
|
Taxa |
Number of specimens (males) |
Percentage of specimens (males) |
||
|
Oise amber |
Baltic amber |
Oise amber |
Baltic amber |
|
|
Leptoconopinae |
2 (1) |
4 (1) |
– |
– |
|
Leptoconops |
2 (1) |
4 (1) |
– |
– |
|
Ceratopogoninae |
7 (4) |
905 (346) |
– |
82.05 (38.23) |
|
Brachypogon |
4( 4) |
158 (44) |
– |
14.32 (27.85) |
|
Stilobezzia |
2 (0) |
29 (10) |
– |
– |
|
Eosphaeromias |
1 (0) |
– |
– |
– |
|
Forcipomyiinae |
74 (5) |
194 (64) |
89.16 (6.76) |
17.59 (32.99) |
|
Forcipomyia |
71 (2) |
175 (57) |
85.54 (2.81) |
15.87 (32.57) |
|
Dasyhelea |
3 (3) |
18 (8) |
– |
– |
|
Total |
83 (10) |
1103 (412) |
– |
– |
It is worth noting here that in the Dominican amber an evidence of mass appearance of another ceratopogonid species was recorded. One piece of amber contained 26 males and 29 females of aquatic Brachypogon dominicanus Szadziewski & Grogan, 1998a, which shows that they were trapped in resin during or after swarming and/or mating activity (Szadziewski and Grogan 1998a, b; Szadziewski and Sontag 2022).
The ceratopogonids described here shed new light on the paleoenvironment in the Ypresian of Oise. The amber bearing forests have been inferred in a fluvio-lacustrine setting without apparent marine influence (Nel et al. 2004). The results of our study reveal marine influence documented by presence of two specimens of the extant, relictual and pantropical genus Leptoconops, which is a good indicator of sea shore or estuarine environments. This record indicates that coastal or estuarine ecosystems close to Oise amber forests were present, which is a new information for the reconstruction of this palaeoenvironment. Indeed, some outcrops of the Oise amber lay at the Eocene seashore (according to the map in Brasero et al. 2009: fig. 1).
Immature stages of extant Brachypogon, Stilobezzia, Sphaeromiini (including Eosphaeromias Szadziewski, Santer, Nel, & Krzemińska gen. nov.), and Dasyhelea are aquatic or semiaquatic and live in different types of fresh water bodies. The small number (10 specimens) of aquatic/semiaquatic biting midges would indicate that fresh water habitats were not common in Oise forests and this image is supported by large number of terrestrial Forcipomyia.
Conspicuous differences in assemblages of biting midges in Oise and Baltic amber seem to exist, even on taking into account the unproportionality in numbers of inclusions compared (Table 1). Nevertheless, such comparison is interesting, because it may reflect differences in (i) age, and (ii) climate in these two localities.
(i) While the age of Oise amber has been safely established at the earliest Eocene (Ypresian), the age of Baltic amber is still a matter of discussions, and various opinions cover c. 20 Ma of the Eocene, from early Eocene (Ypresian; Ritzkovski 1997) to late Eocene (Priabonian; Grimaldi and Ross 2017). The main cause of this uncertainty is that deposits of Baltic amber are moved outside their primary Eocene sediments. In this situation comparison of fauna is very important, because finding of the same species (if only one!) in both ambers would indicate also their similar age (insect species are believed to live up to 5 million years; Simpson 1951). Till now no such species were found, including our study.
(ii) The climates in Oise amber forest and in Baltic amber forests differed conspicuously. The Ypresian is known as the episode of highest temperature in the Cenozoic (PETM: Paleocene–Eocene Thermal Maximum), which was an aftermath of the mass eruptions of volcanoes in Greenland (Ziegler 1988; Obst et al. 2015). From that period on the gradual fall of annual temperature was observed. In the Priabonian, which is the discussed latest age of Baltic amber, the climate in Europe was still subtropical but annual temperature was cooler at c. 4ºC (Zachos et al. 2001). This difference might have an impact both on the plant and faunal assemblages.
There is an intriguing question why some fossil genera, characteristic for the Eocene, are absent in European amber from Oise (53 Ma), while these are widely distributed and common in European Baltic amber (c. 45 Ma) (see Table 2; Fig. 11).
Table 2. Occurrence of fossil indicative genera of Ceratopogonidae in Eocene ambers. After Szadziewski (2018) and recent data.
|
Genus |
Baltic amber 45 Ma |
Oise amber 53 Ma |
Fushun amber 53 Ma |
Sakhalin amber 45 Ma |
Indian amber 54 Ma |
|
Eohelea Petrunkevitch, 1957 |
+ |
– |
– |
+ |
+ |
|
Gedanohelea Szadziewski, 1988 |
+ |
– |
+ |
– |
+ |
|
Mantohelea Szadziewski, 1988 |
+ |
– |
+ |
– |
+ |
This difference cannot be explained by various geological ages, as these taxa were reported even in contemporary early Eocene outcrops from other remote regions of Asia (Fushun in China, 50 Ma; Wang et al. 2014) (Stebner et al. 2016); Indian subcontinent (Cambay, 54.6 Ma; Smith et al. 2016) (Stebner et al. 2017), and middle Eocene Sakhalin amber in Russia (45 Ma; Kodrul 1999). On the other hand, a new genus Eosphaeromias Szadziewski, Santer, Nel, & Krzemińska was found only in the Oise amber, and is absent in large numbers of ceratopogonids in other fossil resins of similar age. The plausible explanation of these faunal differences would be that the Oise forests were unique habitats, highly specific and not suitable for some aquatic or semiaquatic biting midges that lived in more northern latitudes of Eocene Europe and in other regions of Asia.
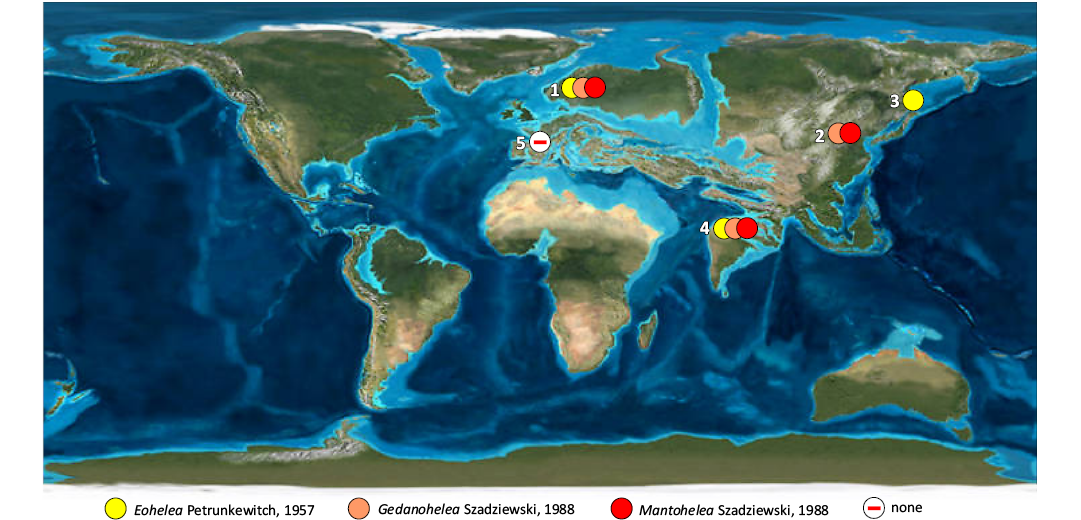
Fig. 11. Distribution of genera of Ceratopogonidae indicative of the lower Eocene (Szadziewski 2018). 1, Baltic amber, 48–34 Ma; Szadziewski (1988); 2, Fushun amber, 50 Ma; Stebner et al. (2016); 3, Sakhalin amber, 45 Ma, Szadziewski (1990); 4, Indian amber, c. 54.6 Ma; Stebner et al. (2017); 5, Oise amber, c. 53 Ma; present study. Map source: Wikimedia Commons. References to amber ages: Baltic (Grimaldi and Ross 2017), Fushun (Wang et al. 2014), Indian (Smith et al. 2016), Oise (Nel et al. 1999); Sakhalin (Kodrul 1999).
Conclusions
In Early Eocene amber from Oise, France the Ceratopogonidae are represented by eight new species in seven genera/subgenera, of which only one genus is extinct and new to science. In comparison with other records from Eocene ambers this sample of examined Ceratopogonidae shows a relatively low diversity. Terrestrial Forcipomyia is the dominant genus (86% of all ceratopogonids, mostly females).
The paleoenvironment of Oise amber forests, as inferred from the assemblage of biting midges, was coastal or close to river estuarium/estuaria. However, absence of some genera known from other Eocene ambers of the world indicates unique forest habitats in the Oise region, more dry and hence less suitable to some aquatic or semiaquatic biting midges.
Acknowledgements
Authors are grateful to Art Borkent (Royal British Columbia Museum, (Victoria, Canada), for his valuable comments on the new genus Eosphaeromias. Jacek Szwedo (University of Gdańsk, Poland) is thanked for help with literature on age of various ambers. This research was supported by the National Science Centre of Poland under Grant No. 2020/37/B/NZ8/03042 to WK. Maxime Santer has benefited from “Ajuts Joan Oró per a la contractació de personal investigador predoctoral en formació (FI-2023), Generalitat de Catalunya”. We also thank Dany Azar (Lebanese University, Beirut, Lebanon) and the anonymous referee for valuable comments.
Editor: Krzysztof Hryniewicz
References
Álvarez-Parra, S., Pérez-de la Fuente, R., Peñalver, E., Barrón, E., Alcalá, L., Pérez-Cano, J., Martín-Closas, C., Trabelsi, K., Meléndez, N., López Del Valle, R., Lozano, R. P., Peris, D., Rodrigo, A., Sarto i Monteys, V., Bueno-Cebollada, C.A., Menor-Salván, C., Philippe, M., Sánchez-García, A., Peña-Kairath, C., Arillo et al. 2021. Dinosaur bonebed amber from an original swamp forest soil. eLife 10: e72477. Crossref
Borkent, A. 2014. The pupae of the biting midges of the World (Diptera: Ceratopogonidae), with a generic key and analysis of the phylogenetic relationships between genera. Zootaxa 3879: 1–327. Crossref
Borkent, A. 2017. 34. Ceratopogonidae (Biting Midges). In: A.H. Kirk-Spriggs and B.J. Sinclair (eds.), Manual of Afrotropical Diptera. Volume 2. Nematocerous Diptera and lower Brachycera. Suricata 5, 733–812. South African National Biodiversity Institute, Pretoria.
Borkent, A. and Dominiak, P. 2020. Catalog of the biting midges of the World (Diptera: Ceratopogonidae). Zootaxa 4787: 1–377. Crossref
Borkent, A., Coram, R.A., and Jarzembowski, E.A. 2013. The oldest fossil biting midge (Diptera: Ceratopogonidae) from the Purbeck Limestone Group (Lower Cretaceous) of southern Great Britain. Polish Journal of Entomology 82: 273–279.
Brasero N., Nel A., and Michez, D. 2009. Insects from the Early Eocene amber of Oise (France): diversity and palaeontological significance. Denisia 26 (86): 41–52.
Clastrier, J. 1974. Leptoconops (Proleptoconops) hutsoni n. sg., n. sp. du Sahara Algerien Septentrional (Diptera, Ceratopogonidae). Parassitologia 16: 231–238.
Crampton, G.C. 1925. A phylogenetic study of the thoracic sclerites of the non-tipuloid Nematocerous Diptera. Annals of the Entomological Society of America 18: 49–69. Crossref
Curtis, J. 1829. British Entomology; Being Illustrations and Descriptions of the Genera of Insects Found in Great Britain and Ireland: Containing Coloured Figures from Nature of the Most Rare and Beautiful Species and in Many Instances of the Plants Upon Which They Are Found. Vol. 6, pls. 242–289. Privately published, London.
Das Gupta, S.K. and Wirth, W.W. 1968. Revision of the Oriental species Stilobezzia Kieffer (Diptera, Ceratopogonidae). United States National Museum Bulletin 283: 1–149. Crossref
Das Gupta, S.K. and Wirth, W.W. 1971. A new genus of Sphaeromiini (Diptera: Ceratopogonidae) from the Oriental Region. Pacific Insects 12: 875–882.
Debenham, M.L. 1970. Australasian Ceratopogonidae (Diptera, Nematocera). Part XII: The status of the genus Heteromyia Say in the Australian Region. Proceedings of the Linnean Society of New South Wales 94: 139–144.
de Franceschi, D. and de Ploëg, G. 2003. Origine de l’ambre des faciès sparnaciens (Éocène inférieur) du Bassin de Paris: le bois de l’arbre producteur. Geodiversitas 25: 633–647.
Dominiak, P. 2012. Biting midges of the genus Dasyhelea Kieffer (Diptera: Ceratopogonidae) in Poland. Polish Journal of Entomology 81: 211–304. Crossref
Dominiak, P. and Szadziewski, R. 2010. Distribution and new synonymy in European biting midges of the genus Dasyhelea Kieffer (Diptera: Ceratopogonidae). Zootaxa 2437: 1–37. Crossref
Eberth, D. and Hamblin, A.P. 1993.Tectonic, stratigraphic, and sedimentologic significance of a regional discontinuity in the upper Judith River Group (Belly River wedge) of southern Alberta, Saskatchewan, and northern Montana. Canadian Journal of Earth Sciences 30: 174–200. Crossref
Goetghebuer, M. 1920. Ceratopogoninae de Belgique. Mémoires du Musée Royal d’Histoire Naturelle de Belgique 8 (3): 1–116. Crossref
Grimaldi, D.A. and Ross, A. 2017. Extraordinary Lagerstätten in amber, with particular reference to the Cretaceous of Burma. In: N.C. Frase and H.-D. Sues (eds.), Terrestrial Conservation Lagerstätten: Windows into the Evolution of Life on Land, 287–342. Dunedin Academic Press Ltd, Edinburgh. Crossref
Iturralde-Vinent, M.A. and MacPhee, R.D.E. 1996. Age and paleogeographical origin of Dominican amber. Science 273: 1850–1852. Crossref
Kieffer, J.J. 1899. Description d’un nouveau genre et tableau des genres européens de la famille des Chironomides (Dipt.). Bulletin de la Société Entomologique de France 1899: 66–70. Crossref
Kieffer, J.J. 1911a. Description de nouveaux chironomides de l’Indian Museum de Calcutta. Records of the Indian Museum 6 : 113–177. Crossref
Kieffer, J.J. 1911b. Nouvelles descriptions de chironomides obtenus d’éclosion. Bulletin de la Societe d’Histoire Naturelle de Moselle (Metz) 27: 1–60.
Kieffer, J.J. 1917a. Chironomides d’Australie conservés au Musée National Hongrois de Budapest. Annales Historico-Naturales Musei Nationalis Hungarici 15: 175–228.
Kieffer, J.J. 1917b. Chironomides d’Amérique conservés au Musée National Hongrois de Budapest. Annales Historico-Naturales Musei Nationalis Hungarici 15: 292–364.
Kieffer, J.J. 1918. Chironomides d’Afrique et d’Asie conservés au Museum National Hongrois de Budapest. Annales Historico-Naturales Musei Nationalis Hungarici 16: 31–136.
Kodrul, T.M. 1999. Paleogene phytostratygraphy of the South Sakhalin. Trudy Geologičeskogo Instituta Rossijskoj Akademii Nauk 519: 1–150.
Kosar C., Green, C., and Pallis, T. 2024. New Jersey amber. In: New Jersey Geological and Water Survey Open-File Report 24-1, 1–8. New Jersey Department of Environmental Protection, New Jersey
Krzemiński, W., Blagoderov, V., Azar, D., Lukashevich, E., Szadziewski, R., Wedmann, S., Nel, A., Collomb, F.M., Waller, A., and Nicholson, D.B. 2019. True flies (Insecta: Diptera) from the late Eocene insect limestone (Bembridge Marls) of the Isle of Wight, England, UK. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 110: 495–554. Crossref
Lenz, F. 1934. B. Die Metamorphose der Heleidae. Heleidae (Ceratopogonidae). 13a. In: E. Lindner (ed.), Die Fliegen der palaearktischen Region, 95–133. E. Schweizerbart’sche Verlagsbuchhandlung, Stuttgart.
Linnaeus, C. 1758. Systema naturae per regna tria naturae, secundum classes, ordines, genera, species, cum caracteribus, differentiis, synonymis, locis. Tomus I. Editio decima, reformata. 824 pp. L. Salvii, Holmiae [= Stockholm]. Crossref
Linnaeus, C. 1767. Systema naturae per regna tria naturae: secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Ed. 12 (rev.), 2 (2), 533–1327. Laurentii Salvii, Holmiae [= Stockholm]. Crossref
Macfie, J.W.S. 1934. Report on
a collection of Ceratopogonidae from Malaya. Annals
of Tropical Medicine and Parasitology 28: 177–194,
279–293. Crossref
Maksoud, S. and Azar, D. 2020. Lebanese amber: latest updates. Palaeoentomology 3: 125–155. Crossref
Meigen, J.W. 1818. Systematische Beschreibung der bekannten europäischen zweiflügeligen Insekten. Erster Theil. xxxvi + 332 pp. F.W. Forstmann, Aachen. Crossref
Newman, E. 1834. Attempted division of British insects into natural orders. Entomological Magazine 2: 379–431.
Nel, A. and Brasero, N. 2010. Oise amber. In: D. Penney (ed.), Biodiversity of Fossils in Amber from the Major World Deposits, 137–148. Siri Scientific Press, Manchester.
Nel, A., de Ploëg, G., Dejax, J., Dutheil, D., de Franceschi, D., Gheerbrant, E., Godinot, M., Hervet, S., Menier, J.J., Augé, M., Bignot, G., Cavagnetto, C., Duffaud, S., Gaudant, J., Hua, S., Jpssang, A., de Lapparent de Broin, F., Pozzi, J.-P., Paicheler, J.-C., Beuchet, F., and Rage, J.-C. 1999. Un gisement sparnacien exceptionnel à plantes, arthropodes et vertébrés (Éocène basal, MP7): le Quesnoy (Oise, France). Comptes rendus de l’Académie des Sciences II A 329: 65–72. Crossref
Nel, A., de Ploëg, G., Millet, J., Menier, J.J., and Waller, A. 2004. The French ambers: a general conspectus and the Lowermost Eocene amber deposit of Le Quesnoy in the Paris Basin. Geologica Acta 2: 3–8. Crossref
Noè, G. 1907. Due nuove specie di ditteri appartenenti ad un genere nuovo. Archivio Zoologico Italiano: Pubblicato Sotto gli Auspicii Della Unione Zoologica 3: 101–163.
Nohra, Y.A., Perrichot, V., Jeanneau, L., Le Polles, L., and Azar, D. 2015. Chemical characterization and botanical origin of French ambers. Journal of Natural Products 78: 1284–1293. Crossref
Obst, K., Ansorge, J., Matting, S., and Hüneke, H. 2015. Early Eocene volcanic ashes on Greifswalder Oie and their depositional environment, with an overview of coeval ash-bearing deposits in northern Germany and Denmark. International Journal of Earth Sciences 104: 2179–2212. Crossref
Ogg, J.G., Ogg, G., Grandstein, F.M. 2008. The Concise Geologic Time Scale. 177 pp. Cambridge University Press, Cambridge.
Petrunkevitch, A. 1957. Eohelea stridulans, gen. nov., sp. nov., a striking example of paramorphism in an amber biting-midge. Journal of Paleontology 31: 208–214.
Ritzkovski, S. 1997. K-Ar Alterbestimmungen der bernsteinführenden Sediment des Samlandes (Paläogen, bezirk Kaliningrad). Metalla 66: 19–23.
Ross, A. and Self, A.E. 2014. The fauna and flora of the Insect Limestone (late Eocene), Isle of Wight, UK: Introduction, history and geology. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 104: 233–244. Crossref
Saint Martin, J.-P., Dutour, Y., Ebbo, L., Frau, C., Mazière, B., Néraudeau, D., Saint Martin, S., Tortosa, T., Turini, E., and Valentin, X. 2021. Reassessment of amber-bearing deposits of Provence, southeastern France. BSGF-Earth Sciences Bulletin 192 (5): 1–22. Crossref
Saunders, L.G. 1957. Revision of the genus Forcipomyia based on characters of all stages (Diptera, Ceratopogonidae). Canadian Journal of Zoology 34: 657–705 [for 1956]. Crossref
Simpson, G.G. 1951. The species concept. Evolution 5: 285–298. Crossref
Skuse, F.A.A. 1889. Diptera of Australia. Part VI.—The Chironomidae. Proceedings of the Linnean Society of New South Wales 4: 215–311. Crossref
Smith, T., Kumar, K., Rana, R.S., Folie. A., Sole´, F., Noiret, C., Steeman, T., Sahni, A., and Rose K.D. 2016. New early Eocene vertebrate assemblage from western India reveals a mixed fauna of European and Gondwana affinities. Geoscience Frontiers 7: 969–1001. Crossref
Solórzano Kraemer, M.M., Delclòs, X., Clapham, M.E., Arillo, A., Peris, D., Jäger P., Stebnerg, F., and Peñalver, E. 2018. Arthropods in modern resins reveal if amber accurately recorded forest arthropod communities. PNAS 115: 6739–6744. Crossref
Sontag, E. and Szadziewski, R. 2011. Biting midges (Diptera: Ceratopogonidae) in Eocene Baltic amber from the Rovno region (Ukraine). Polish Journal of Entomology 80: 779–800. Crossref
Spinnelli, G.R. 1997. A new genus of the tribe Sphaeromiini from subantarctic Argentina and Chile related to Mackerrasomyia Debenham (Diptera: Ceratopogonidae). Memoirs of the Entomological Society of Washington 18: 224–229.
Stebner, F., Szadziewski, R., and Wang, B. 2016. Biting midges (Diptera: Ceratopogonidae) in Fushun amber reveal further biotic links between Asia and Europe during the Eocene. Palaeontologia Electronica 19.3.31A: 1–9. Crossref
Stebner, F., Szadziewski, R., Singh, H., Gunkel, S., and Rust, J. 2017. Biting midges (Diptera Ceratopogonidae) from Cambay amber indicate that the Eocene fauna of the Indian subcontinent was not isolated. PLOS ONE 12 (1) e0169144. Crossref
Szadziewski, R. 1988. Biting midges (Diptera, Ceratopogonidae) from Baltic amber. Polskie Pismo Entomologiczne 58: 3–283.
Szadziewski, R. 1990. Biting midges (Insecta: Diptera: Ceratopogonidae) from Sakhalin amber. Prace Muzeum Ziemi 41: 77–81.
Szadziewski R. 1996. Biting midges from Lower Cretaceous amber of Lebanon and Upper Cretaceous Siberian amber of Taimyr (Diptera, Ceratopogonidae). Studia Dipterologica 3: 23–86.
Szadziewski, R. 2005. The first fossil species in the predaceous tribe Sphaeromiini (Diptera: Ceratopogonidae). Polskie Pismo Entomologiczne 74: 363–368.
Szadziewski, R. 2018. Biting midges (Diptera: Ceratopogonidae) as indicators of biostratigraphy, ecological reconstructions and identification of amber deposits. Earth and Environmental Science Transactions of the Royal Society of Edinburgh 107: 219–230. Crossref
Szadziewski, R. and Grogan, W.L. 1996. Biting midges (Diptera: Ceratopogonidae) from Mexican amber. Polish Journal of Entomology 65: 291–295.
Szadziewski, R. and Grogan, W.L. 1998a. Biting midges from Dominican amber III. Species of the tribes Culicoidini and Ceratopogonini (Diptera: Ceratopogonidae). Insecta Mundi 12: 39–49.
Szadziewski, R. and Grogan, W.L. Jr. 1998b. Biting midges from Dominican amber. IV. Species of the tribes Dasyheleini and Forcipomyiini (Diptera: Ceratopogonidae). Polskie Pismo Entomologiczne 67: 255–290
Szadziewski, R. and Sontag, E. 2022. Komunikowanie się samców i samic muchówek z rodziny kuczmanów (Diptera: Ceratopogonidae). Kosmos 72: 317–323. Crossref
Szadziewski, R., Arillo, A., Urbanek, A., and Sontag, E. 2016. Biting midges of the extinct genus Protoculicoides Boesel from Lower Cretaceous amber of San Just, Spain and new synonymy in recently described fossil genera (Diptera: Ceratopogonidae). Cretaceous Research 58: 1–9.
Szadziewski, R., Gwizdalska-Kentzer, M., and Sontag, E. 2007. Predatory biting midges of the genus Sphaeromias in Europe (Diptera: Ceratopogonidae). Polish Journal of Entomology 76: 293–302.
Szadziewski, R., Krzywiński, J., and Giłka, W. 1997. Ceratopogonidae. In: A. Nilsson (ed.), Aquatic Insects of North Europe—A Taxonomic Handbook, Vol. 2: Odonata—Diptera, 243–263. Apollo Books, Stenstrup.
Wang, B., Rust, J., Engel, M.S., Szwedo, J., Dutta, S., Nel, A., Fan, Y., Meng, F., Shi, G., Jarzembowski, E.A., Wappler, T., Stebner, F., Fang, Y., Mao, L., Zheng, D., and Zhang, H. 2014. A diverse paleobiota in early Eocene Fushun amber from China. Current Biology 24: 1606–1610. Crossref
Winnertz, J. 1852. Beitrag zur Kenntniss der Gattung Ceratopogon Meigen. Linnaea Entomologica 6: 1–80. Crossref
Wirth, W.W. 1962. A reclassification of the Palpomyia-Bezzia-Macropeza groups and a revision of the North American Sphaeromiini (Diptera, Ceratopogonidae). Annals of the Entomological Society of America 55: 272–287. Crossref
Wirth, W.W. and Blanton, F.S. 1972. Lanehelea, a new Neotropical genus of Sphaeromiini (Diptera: Ceratopogonidae). Studia Entomologica 15: 433–438.
Wirth, W.W. and Grogan, W.L. Jr. 1988. The predaceous midges of the World (Diptera: Ceratopogonidae; Tribe Ceratopogonini). In: Flora and Fauna Handbook, Vol. 4, 1–160. E.J. Brill, New York. Crossref
Zachos, J., Pagani, M., Sloan, L., Thomas, E., and Billups, K. 2001. Trends, rhytms and abberrations in global climate 65 Ma to present. Science 292: 686–693. Crossref
Ziegler, P.A. 1988. Evolution of the Arctic-North Atlantic and Western Tethys. American Association of Petroleum Geologists (Tulsa), Memoir 43: 1–197. Crossref
Acta Palaeontol. Pol. 71 (1): 51–68, 2026
https://doi.org/10.4202/app.01223.2024