

Exceptionally preserved Oligocene emperor butterfly from France provides a new calibration point for Apaturinae evolution
HOSSEIN RAJAEI, TORSTEN WAPPLER, RIENK DE JONG, NIKLAS WAHLBERG, and MICHAEL S. ENGEL
Rajaei, H., Wappler, T., De Jong, R., Wahlberg, N., and Engel, M.S. 2026. Exceptionally preserved Oligocene emperor butterfly from France provides a new calibration point for Apaturinae evolution. Acta Palaeontologica Polonica 71 (1): 185–191.
A new genus and species of emperor butterfly (Nymphalidae, Apaturinae) is described based on an exceptionally well-preserved specimen from the “Calcaire de Campagne Calavon” limestone of southern France (early Oligocene age, approximately 34–28 Mya). Most parts of the right wings and upper parts of the left wings are visible, with complete venation and even wing patterns. The head and thorax are clearly visible from both sides, and most of the abdomen is preserved. Apaturoides monikae gen. et sp. nov., is compared to all genera of the subfamily and based on the form of the wings and venation is considered most similar to species of Apatura, today occurring widely in Palearctic, excluding North Africa and Middle East. This discovery provides an important calibration point based on direct fossil evidence for estimating relationships and divergence estimates among emperor butterflies.
Key words: Lepidoptera, Papilionoidea, Nymphalidae, Apaturinae, new genus, new species, Oligocene, France.
Hossein Rajaei [hossein.rajaei@smns-bw.de; ORCID: https://orcid.org/0000-0002-3940-3734], State Museum of Natural History, Stuttgart, Rosenstein 1-3, 70191 Stuttgart, Germany.
Torsten Wappler [Torsten.Wappler@hlmd.de; ORCID: https://orcid.org/0000-0003-1592-0988], Hessisches Landesmuseum Darmstadt, Friedensplatz 1, 64283 Darmstadt, Germany; Bonn Institute of Organismic Biology (BIOB), Section of Palaeontology, Nussallee 8, 53115 Bonn, Germany.
Rienk de Jong [Rienk.deJong@naturalis.nl; ORCID: https://orcid.org/0000-0002-2052-5993], Department of Entomology, Naturalis Biodiversity Center, PO Box 9517, 2300 RA Leiden, The Netherlands.
Niklas Wahlberg [Niklas.wahlberg@biol.lu.se; ORCID: https://orcid.org/0000-0002-1259-3363], Department of Biology, Lund University, 223 62 Lund, Sweden.
Michael S. Engel [mengel@amnh.org; ORCID: https://orcid.org/0000-0003-3067-077X], Division of Invertebrate Zoology, American Museum of Natural History, Central Park West at 79th Street, New York, NY 10 024, USA.
Received 14 January 2026, accepted 3 February 2026, published online 26 March 2026.
Copyright © 2026 H. Rajaei et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (for details please see http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Butterflies (Lepidoptera: Papilionoidea) are among the most iconic and beloved insects, capturing the human imagination for millennia (Grimaldi and Engel 2005). They are also among the most intensely studied groups of insects, serving as models for everything from community ecology to the molecular basis of pattern development. Yet, in terms of their evolution, butterflies are a comparatively young clade, with hypothesized stem groups arising in the Late Cretaceous but almost entirely diversifying in the Cenozoic (Grimaldi and Engel 2005; Wahlberg et al. 2013; Espeland et al. 2018; Kawahara et al. 2019, 2023). This is in stark contrast to the putative Paleozoic age of Lepidoptera (Kawahara et al. 2019). Direct fossil evidence, however, is limited and much remains speculative. Due to taphonomical processes, fossil occurrences of Lepidoptera are infrequent and most of them have been discovered in amber, thus limiting their records to deposits of the Early Cretaceous or younger age (Martínez-Delclòs and Martinell 1993; Sarto i Monteys et al. 2022). This natural bias leads to considerable discrepancies in divergence-time estimates in molecular analyses (Labandeira and Sepkoski 1993; Kristensen and Skalski 1998; Kozlov et al. 2002; Sohn et al. 2015). Until recently, Archaeolepis mane Whalley, 1985, represented the oldest fossil occurrence for the Lepidoptera, dating back to the Sinemurian (Early Jurassic, ca. 190 Mya, from Dorset, England) (Whalley 1985). However, subsequent discovery of fossilized wing scales from sediments of the latest Triassic and earliest Jurassic age in northern Germany confirmed the presence of not only Lepidoptera, but also the clade Coelolepida (including Glossata), pushing age estimates for the lineage back to at least 200 Mya (Van Eldijk et al. 2018). Recently, Fiorelli et al. (2025) reported hexapod scales dating to 236 Mya, which they interpreted as belonging to the clade Glossata. Other lepidopteran fossils have been summarized by Sohn et al. (2012, 2015).
As for the butterflies (Superfamily Papilionoidea), Sohn et al. (2012, 2015) reported 142 fossil occurrences (Sohn et al. 2012, 2015), which potentially represents the richest fossil record for any superfamily across the Apoditrysia (including Obtectomera and Macrolepidoptera) and the second richest fossil record for a superfamily in the Ditrysia, after Tineoidea (Sohn et al. 2015). However, a revision of the butterfly fossil record by Jong (2017) suggests that only 48 of these are butterflies, and the remaining fossils either cannot be confidently assigned to Papilionoidea, or were wrongly identified in the original description. Of the 48 validated fossil butterflies, almost half (23) are assigned to the family Nymphalidae (Jong 2017). Two of the fossil species are particularly striking with preserved wing patterns, namely Prodryas persephone Scudder, 1878, of the Nymphalinae Rafinesque, 1815, from the Eocene/Oligocene boundary of Florissant, Colorado, USA, and Dynamine alexae Peñalver & Grimaldi, 2006, of the subfamily Biblidinae Boisduval, 1833, from the Early Miocene Dominican amber (ca. 15–17 Mya) (Grimaldi and Engel 2005; Peñalver and Grimaldi 2006). Recently, Fischer and Hausmann (2025) reported the first reliable fossil record of a butterfly (Papilionoidea) in Baltic amber, based on the discovery of a characteristic hexagonally sculptured egg that can be attributed to the admiral subfamily Limenitidinae (Nymphalidae). This finding establishes a minimum Eocene age (33.9–37.8 Mya) for this group and provides a rare direct calibration point in the evolutionary history of butterflies.
With over 80 described species in 22 genera, the subfamily Apaturinae Boisduval, 1840, is mainly distributed in the Palearctic, Indo-Australian, Afrotropical (Apaturopsis) and New World (Doxocopa and Asterocampa) regions (Masui et al. 2011). Until now, no fossils have been found that can be definitively ascribed to the Apaturinae (Sohn et al. 2012; Jong 2017). Here we report the first fossil for the subfamily Apaturinae. The fossil provides a new biogeographic and temporal calibration point for understanding the evolutionary history and diversification of butterflies.
Institutional abbreviations.—NHMMZ, Landessammlung für Naturkunde Rheinland-Pfalz at the Naturhistorisches Museum Mainz, Germany.
Other abbreviations.—A, anal vein; CuA, cubital-anterioris vein; M, medial vein; R, radial vein; Sc, subcostal vein.
Nomenclatural acts.— This published work and the nomenclatural acts it contains have been registered in ZooBank: urn:lsid:zoobank.org:pub:2CFB7834-82E0-4B78-A3C7-2818C32D72CC
Material and methods
The specimen reported here is from the lower Oligocene lacustrine laminate exposed along the northern flank of the Montagne de Lubéron, Alpes-de-Haute-Provence, south of the village of Céreste. Today, these sites that contain fossils are protected by the Lubéron National Geological Nature Reserve (Coster and Legal 2021). The fossils were found in clay-limestone laminates (also called Papierschiefer), which belong to the lacustrine Campagne-Calavon Formation. This formation is dated as Rupelian (biozone MP23–24, 33.9–27.82 Mya) based on biostratigraphic data from gastropods (Cavelier et al. 1984), charophytes (Feist 1977), ostracods (Apostolescu and Guernet 1992), and mammals (Helmer and Vianey-Liaud 1970; Ducreux et al. 1985). The geologic history of the Campagne-Calavon Formation is marked by the transformation of an environment that was once characterized by calm and shallow lakes, with fluctuating salt levels over time (Ducreux 1982; Lutz 1984), and bears numerous fossil insects, mammals, birds, fish, and plant remains (e.g., Lutz 1984, 1985a, b; Mayr 2000; Nel et al. 2024; Pfretzschner 1998; Nidergas et al. 2018; Heie and Lutz 2002; Wappler 2004; Claisse et al. 2019).
The type specimen (NHMMZ PE 2019/1-LS) is deposited in NHMMZ.
The fossil is preserved between two asymmetrical rhombus-shaped limestone layers (longest diameter 15.2 × 8.8 cm. No crack is visible on the limestone layer with the print of the upperside (= part), but the layer with underside’s print (= counterpart) has three cracks on the upper corner and one crack on its lower part. The butterfly (Fig. 1) has right wings nearly completely preserved, as well as most of the left forewing. The head (except antennae), thorax, and abdomen are well preserved. The fossil was collected by Herbert Lutz (Seeheim-Jugenheim, Germany) during his excavations in 1979, and who kindly offered us the opportunity to examine and conduct further research.
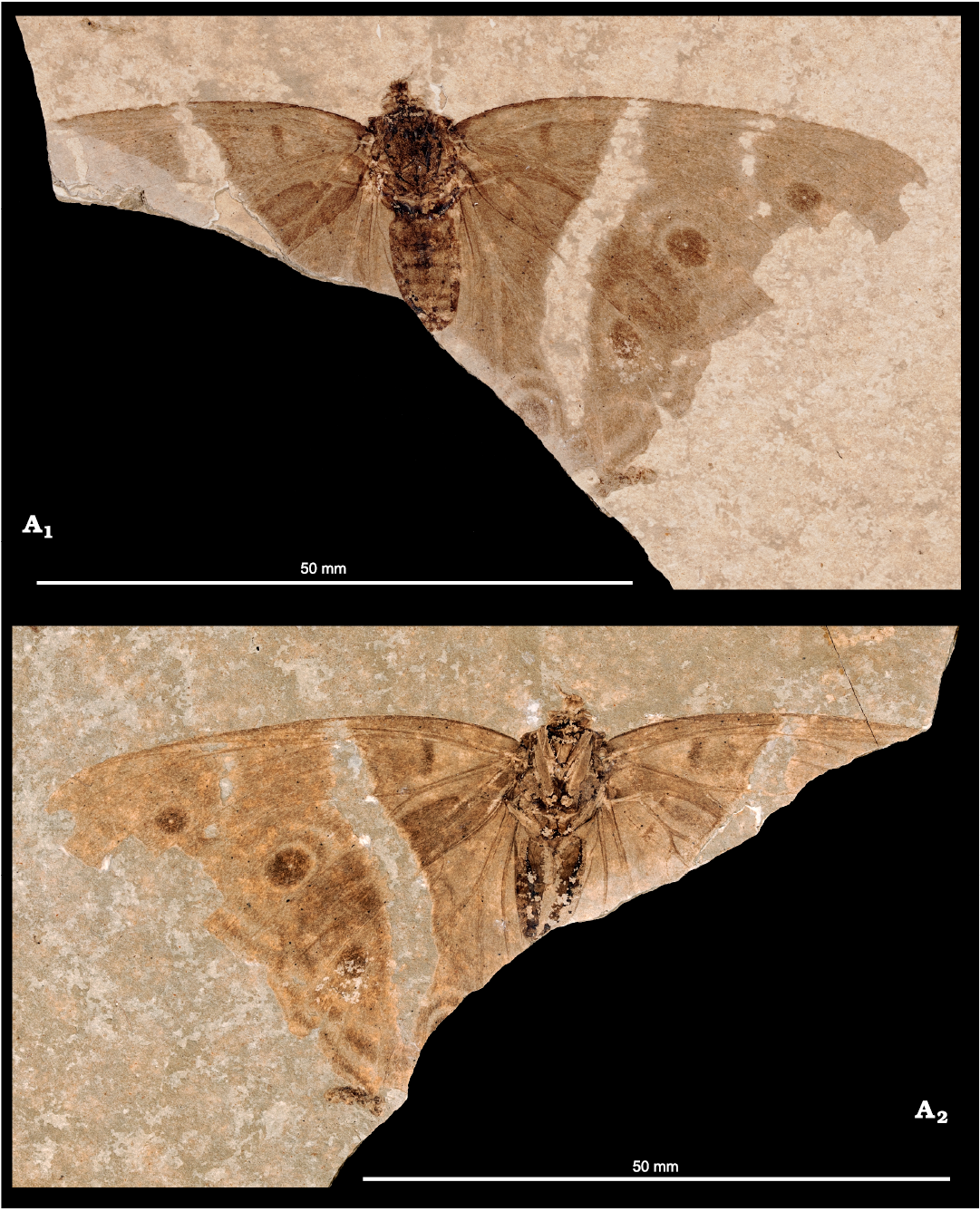
Fig. 1. A nymphalid butterfly Apaturoides monikae gen. et sp. nov. from lower Oligocene lacustrine laminate, Alpes-de-Haute Provence, south of the village of Céreste, France. NHMMZ PE 2019/1-LS, part in dorsal view (A1), counterpart in ventral view (A2).
The fossil was examined using a Leica stereomicroscope (model: M205C). Photographs were taken using a Keyence digital microscope (model: VHX–5000) and processed using Adobe Photoshop CS5. Digital line drawings were made in Adobe Illustrator (version: CS5) based on the photos and the original fossil. The systematics of Nymphalidae follows Espeland et al. (2018) and Wahlberg et al. (2009), and the higher classification follows Nieukerken (2011). For nomenclature of morphological characters and wing venations we follow Ackery et al. (1999).
Systematic paleontology
Superfamily Papilionoidea Latreille, 1802
Family Nymphalidae Rafinesque, 1815
Subfamily Apaturinae Boisduval, 1840
Genus Apaturoides nov.
Zoobank LSID: urn:lsid:zoobank.org:pub:2CFB7834-82E0-4B78-A3C7-2818C32D72CC
Etymology: A combination of Apatura and Ancient Greek eidḗs (ειδής), resembling, connected to, or of the nature of; and is meant to emphasize the close relationship between the genera. The gender is feminine.
Type species: Apaturoides monikae sp. nov.; monotypic, see below.
Diagnosis.—As for the type species by monotypy.
Apaturoides monikae gen. et sp. nov.
Figs. 1, 2.
Zoobank LSID: urn:lsid:zoobank.org:pub:2CFB7834-82E0-4B78-A3C7-2818C32D72CC
Etymology: In honor of Monika Lutz-Scholz on her 70th birthday (born 28 October 1956). Monika is the wife of Herbert Lutz, who discovered the fossil in 1979.
Holotype: NHMMZ PE 2019/1-LS, a well-preserved body with most parts of right wings and small part of the left wings.
Type locality: “Calcaire de Campagne Calavon” limestone of Céreste-en-Luberon (also known as Céreste), a locality situated within the commune of Alpes-de-Haute-Provence, in the Alpes-Maritimes region of south-eastern France (43.9°N, 5.6°E).
Type horizon: Rupelian (dated as 33.9–28.4 Mya), lower Oligocene.
Diagnosis.—The presence of open wing cells is a key character that assigns this new species to Nymphalidae. While this character is not universally found in Nymphalidae, it is exceedingly rare in other papilionoid families (e.g., some Hesperiidae species). However, the new species can be excluded from Hesperiidae based on the shape of head, thorax, wing, and other venational details.
In terms of wing pattern (Figs. 1, 2), the new fossil bears a strong resemblance to species of Junonia (Nymphalinae), particularly to Junonia orithya (Linnaeus, 1758), which has quite similar eye spots. However, in species of Junonia the origin of R1 in the forewing is generally closer to the wing base than in the fossil, and the origin of CuA2 is definitely closer to CuA1.
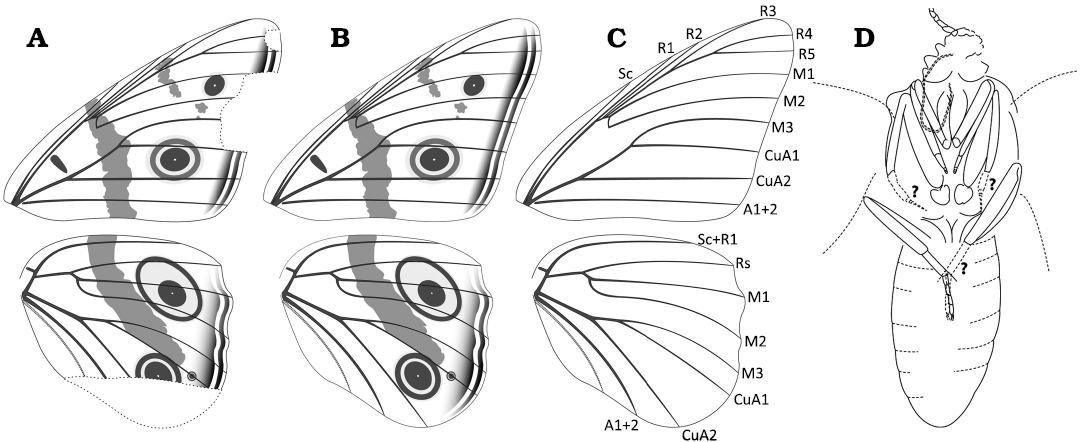
Fig. 2. Wing pattern, venation and details of the ventral side of the body of nymphalid butterfly Apaturoides monikae gen et sp. nov. A. Digital drawing of the wing pattern. B. Reconstruction of the wing pattern, including missing portions. C. Wing venation. D. Line drawing of the head, thorax, and abdomen, as far as visible on the fossil. Abbreviations: A, anal vein; CuA, cubital-anterioris vein; M, medial vein; R, radial vein; Sc, subcostal vein.
Comparing the wing shape and venation with representatives from all twelve subfamilies of Nymphalidae allows for a more accurate identification. The wing shape of the new species shows some deviation from many nymphalids; for example, the forewing is relatively elongate, and the angle between its inner and outer margins is about 73°, which is uncommon for this family. Such a wing shape can be found in some subfamilies, such as Limenitidinae (Neurosigma); Biblidinae (Batesia), Danainae (Danaus), and Satyrinae (Zethera and Penthema). However, in all of these examples, the cell is closed and the origin of CuA2 in the forewing is closer to the wing base rather than to the origin of CuA1, whereas in the new species, the cell is open and CuA2 originates roughly at the middle of the wing base and CuA1. These characters confidentially assign the new species to the subfamily Apaturinae.
We thus compared the new species with representatives of all known genera within the subfamily Apaturinae. Currently, 22 genera are recognized in Apaturinae. Among them, one genus (Apaturopsis) is confined to Africa, another (Asterocampa) is found only in North America, one (Doxocopa) occurs in both North and South America, and the remaining genera are distributed in the Palearctic-Oriental regions. The new species differs from all species of first three genera, as in all of them the origin of CuA2 is distinctly closer to the origin of CuA1 than to the wing base. In the other genera, with some variation, the origin of CuA2 is closer to CuA1. Among all other genera, only Apatura has a somewhat similar pattern to the new species, where CuA2 originates close to the middle of the wing base. Despite these similarities, the studied specimen does not belong to this genus, as Apatura has a different wing shape, with a much broader angle (almost 90°) between the inner and outer margins of the forewing, as well as a different shape of the hindwing.
Description.—Head: Eyes only partially visible; right antenna with only four basal preserved antennomeres, left antenna missing. Haustellum well-developed, ca. 0.9 mm in length, uncoiled (Figs. 1A2, 2D).
Thorax: Scaled. Legs as far as visible well-developed; forelegs not notably reduced; only right metatarsus visible.
Wings:
Wingspan ca. 90 mm, forewing length ca. 41 mm, with rounded apex.
Forewing margins smooth; two eyespots, upper one located between M1 and
M2, formed of a dark spot with white dot in center and a
lighter outer ring, max. diameter of dark spot 3.1 mm; lower
eyespot located between veins CuA1 and CuA2, formed of a dark
spot with a white dot in its center, max. diameter of dark
spot 4.3 mm, two light outer rings separated by a darker
ring; shadow of postmedial and subterminal lines scarcely visible in
forewing; a streak smear present in basal area, a grey
band in the middle area as well a shorter thin band in
subcostal area of forewing seem to be the preserved wing
patterns. Hindwing termen waved on M; three eyespots visible: upper one
located between Rs and M2 and formed of a dark spot with a
white dot centrally and a lighter outer ring,
maximum diameter of dark spot 3.7 mm; smallest eyespot located on CuA1
and formed of a dark spot with a white dot
centrally, max. diameter of dark spot 1.1 mm, and has a light
outer ring; lower eyespot located between CuA1 and CuA2, formed of a
dark spot with a white dot centrally, max. diameter
of dark spot 3.9 mm, two light outer rings separated by a
darker ring; shadow of postmedial and subterminal lines scarcely
visible on hindwing, same as the forewings preserved wing patterns are
visible as a grey band on the middle of costal area of
hindwing and continuing to CuA1. Venation of both wings
well visible. Forewing (Figs. 1A, 2A–C) with an open cell; radial veins
not swollen; R1 and R2 originating from cell; R3–5 stalked together;
M1 originates from end of cell; M2 ends at halfway between M1 and M3; M3
and CuA1 stalked together; CuA2 originates at halfway between wing base
and CuA1; A1+2 originate from wing base. Hindwing (Figs. 1A, 2A–C) has
an open cell; Sc+R1 originates from wing base; Rs, M1, and M2 originate
from cell; M2 ends halfway between M1 and M3; M3 and CuA1 stalked
together; A1+2 originates from wing base (hindwings close to abdomen
such that A3 is covered by the abdomen).
Abdomen: Relatively short, thick; its dorsal end partially absent.
Remarks.—Identifying fossil Lepidoptera at lower taxonomic levels is often challenging because the necessary apomorphic characters for correct identification are rarely well-preserved, such as the three longitudinal ridges on the ventromesal surface of the antennae, an apomorphy of the Nymphalidae. Even assignment of such fossils to higher taxonomic levels may be problematic. However, relying on combinations of visible characters on the current fossil can be helpful in family and tribal assignment of this species. We consider structural characters such as wing venation better indicators for phylogenetic relationships rather than wing pattern, as elements such as eye spots have apparently evolved independently in different subfamilies (Hanotte et al. in press.).
Stratigraphic and geographic range.—Rupelian (33.9–28.4 MYR BP), middle Oligocene; Céreste, between the Vaucluse and the Alpes-de-Haute-Provence, France.
Concluding remarks
Given the likely relationship with Apatura, the fossil could be ascribed to an Old-World group within the Apatura-clade (Wahlberg et al. 2009; Freitas and Brown 2004; Ohshima et al. 2010). Based on molecular estimates, the subfamily diverged from Biblidinae, approximately 47–52 Mya (Chazot et al. 2019; Kawahara et al. 2023). However, it only began to diversify into the modern genera around 33 Mya (Chazot et al. 2019; Kawahara et al. 2023), which is roughly near to the age of the current fossil. Around this time, the two major clades within Apaturinae, the Apatura-clade and the Chitoria-clade, diverged from each other. The events that transpired during the intervening period remain largely unknown, possibly due to large-scale extinctions, of which this fossil seems to be an example. Initially, the subfamily diversified in SE Asia, subsequently dispersing to Africa once around 21 Mya (Apaturopsis) and to North America at least twice, approximately 17 Mya (Doxocopa) and 16 Mya (Asterocampa), respectively (Chazot et al. 2021). The subfamily continued to proliferate in Asia, leading to the majority of the modern diversity in this region.
The Apatura-clade itself, comprising the genera Apatura, Hestina, Mimathyma, Euripus, Sephisa, Hestinalis, and Doxocopa, began diversifying around 18 Mya, based on current molecular estimates (Chazot et al. 2019; Kawahara et al. 2023), suggesting that the fossil would represent an extinct stem lineage of the Apatura-clade. The same molecular estimates place the divergence of Apatura from its sister lineage to about 16 Mya, suggesting that either molecular estimates are too young and the Apatura lineage dates to at least 34–28 Mya, or extant Apatura retain ancestral morphological features. Extant Apatura comprises four species that are distributed throughout the temperate Palaearctic region, and are closely related to each other having diversified in the past 8 million years or so (Chazot et al. 2021).
Relationships of the genera, including Apatura, within the Apatura-clade are somewhat unstable. The subfamily has been the focus of only one phylogenetic study (Ohshima et al. 2010), but a similar number of taxa have been included in the recent broader butterfly studies (Chazot et al. 2019, 2021; Kawahara et al. 2023). None of these studies show the same relationships of the genera within the Apatura-clade, meaning that further phylogenetic work is needed to resolve the relationships of the extant taxa. In the meantime, the fossil provides a minimum age for the Apatura-clade.
Acknowledgements
The authors would like to thank
Herbert Lutz (Seeheim-Jugenheim, Germany) for drawing their attention
to this important fossil. Furthermore, we
thank the Naturhistorisches Museum Mainz
(Germany)/Landessammlung für Naturkunde Rheinland-Pfalz and its staff
for providing access to fossil material. We
would like to thank the
reviewers, Axel Hausmann (Bavarian State Collection of Zoology,
Munich, Germany) and Vazrick Nazari (University of Padua, Italy), for
their constructive suggestions, which helped to
improve this manuscript.
Editor: Krzysztof Hryniewicz
References
Ackery, P.R., Jong, R. de, and Vane-Wright, R.I. 1999. The butterflies: Hedyloidea, Hesperioidea and Papilionoidea. In: N.P. Kristensen (ed.), Lepidoptera, Moths and Butterflies, Vol. 1, 263–300. W. de Gruyter, Berlin. Crossref
Apostolescu, V. and Guernet, C. 1992. Les ostracodes oligocènes de la région Forcalquier-Manosque (Bassin continental d’Apt, Haute-Provence). Revue de Micropaléontologie 35: 91–115.
Cavelier, C., Alabouvette, B., Amberger, G., Cautru, J.-P., Charolais, J., Chateauneuf, J.-J., Crochet, J.-Y., Campredon, R., Debeglia, N., Durand, J.-P., Feist-Castel, M., Freytet, P., Gannat, E., Gaudant, J., Giannerini, G., Godinot, M., Hartenberger, J.-L., Hugueney, M., Kerckhove, C., Lefavrais-Raymond, A., Legendre, S., Lespinasse, P., Magne, J., Nury, D., Pairis, J.-L., Plaziat, J.-C., Remy, J.-A., Schlund, J.-M., Sige, B., Sittler, C., Sudre, J., Trauth, N., Truc, G., Valleron, M.-M., Vianey-Liaud, M., and Villate, J. 1984. Paléogène. In: S. Debrand-Passard, S. Courbouleixand, and M.-J. Liehardt (eds.), Synthèse géologique du Sud-Est de la France, 389–468. BRGM, Orléans.
Chazot, N., Condamine, F., Dudas, G., Peña, C., Kodandaramaiah, U., Matos-Maraví, P., Aduse-Poku, K., Elias, M., Warren, A.D., Lohman, D., Penz, C., DeVries, P., Fric, Z. F., Nylin, S., Müller, C., Kawahara, A.Y., Silva-Brandao, K., Lamas, G., Kleckova, I., Zubek, A., Ortiz-Acevedo, E., Vila, R., Vane-Wright, R., Mullen, S., Jiggins, C.D., Wheat, C.W., Freitas, A.V.L., and Wahlberg, N. 2021. Conserved ancestral tropical niche but different continental histories explain the latitudinal diversity gradient in brush-footed butterflies. Nature Communications 12: 5717. Crossref
Chazot, N., Wahlberg, N., Freitas, A.V.L., Mitter, C., Labandeira, C., Sohn, J.-C., Sahoo, R. K., Seraphim, N., Jong, R. de, and Heikkilä, M. 2019. Priors and posteriors in Bayesian timing of divergence analyses: the age of butterflies revisited. Systematic Biology 68: 797–813. Crossref
Claisse, P., Brisac, P., and Nel, A. 2019. The first fossil Nemopteridae from the Oligocene of Céreste (France) (Insecta: Neuroptera). Palaeoentomology 2 (2): 115–118. Crossref
Coster, P. and Legal, S. 2021. An Early Oligocene fossil Lagerstätten from the lacustrine deposits of the Luberon UNESCO Global Geopark. Geoconservation Research 4: 1–9.
Ducreux, J.L. 1982. Recherches de Concentrations Métallifères en Domaine paralique et limnique—le cas des Formations du Crétacé Supérieur gardois, de l’Oligocène de Manosque (Alpes de Haute Provence) et de St. Martin des Plans (Puy de Döme). 342 pp. Ph.D. thesis, Université Claude Bernard, Lyon.
Ducreux, J.L., Hugueney, M., and Truc, G. 1985. La formation des calcaires et lignites de Sigonce (Oligocène moyen, Bassin de Forcalquier, Alpes de Haute-Provence): datation à l’aide des mammifères; reconstitution des milieux de dépôts. Geobios 18: 109–114. Crossref
Espeland, M., Breinholt, J., Willmott, K.R., Warren, A.D., Vila, R., Toussaint, E.F.A., Maunsell, S.C., Aduse-Poku, K., Talavera, G., Eastwood, R., Jarzyna, M.A., Guralnick, R., Lohman, D.J., Pierce, N.E., and Kawahara, A.Y. 2018. A comprehensive and dated phylogenomic analysis of butterflies. Current Biology 28: 770–77. Crossref
Feist, M. 1977. Étude floristique et biostratigraphique des Charophytes dans les séries du Paléogène de Provence. Géologie Méditerranéenne 4: 109–138. Crossref
Fiorelli, L.E., Vera, E.I., Perez Loinaze V.S., Torréns, J., Ezcurra, M.D., Lara, M.B., and Desojo J.B. 2025. Back to the poop: the oldest hexapod scales discovered within a Triassic coprolite from Argentina. Journal of South American Earth Sciences 162: 105584. Crossref
Fischer, T.C. and Hausmann, A. 2025. First butterfly (Papilionoidea) from Baltic amber by a characteristic egg pinpoints an Eocene minimal age of admirals (Nymphalidae: Limenitidinae)—a distinct step in the rise of the Papilionoidea. Palaeontologia Electronica 28 (1): a10. Crossref
Freitas, A.V.L. and Brown, K.S., Jr. 2004. Phylogeny of the Nymphalidae (Lepidoptera). Systematic Biology 53: 363–383. Crossref
Grimaldi, D. and Engel, M.S. 2005. Evolution of the Insects. xv + 755 pp. Cambridge University Press, Cambridge.
Hanotte, B., Willink, B., & Monteiro, A. (2024). Eyespots originated multiple times independently across the Lepidoptera. bioRxiv [published online https://doi.org/10.1101/2024.02.07.579046] Crossref
Heie, O.E. and Lutz, H. 2002. Fossil aphids from the Early Oligocene deposits near Céreste, France, with descriptions of new genera and species (Hemiptera, Sternorrhyncha, Aphidoidea). Mainzer Naturwissenschaftliches Archiv 40: 113–122.
Helmer D. and Vianey-Liaud, M. 1970. Nouveaux gisements de rongeurs dans l’Oligocène moyen de Provence. Compte Rendu Sommaire des Séances de la Société Géologique de France 2: 45–46.
Jong, R. de 2017. Fossil butterflies, calibration points and the molecular clock (Lepidoptera: Papilionoidea). Zootaxa 4270: 1–63. Crossref
Kawahara, A.Y., Plotkin, D., Espeland, M., Meusemann, K., Toussaint, E.F.A., Donath, A., Gimnich, F., Frandsen, P.B., Zwick, A., dos Reis, M., Barber, J.R., Peters, R.S., Liu, S., Zhou, X., Mayer, C., Podsiadlowski, L., Storer, C., Yack, J.E., Misof, B., and Breinholt, J.W. 2019. Phylogenomics reveals the evolutionary timing and pattern of butterflies and moths. Proceedings of the National Academy of Sciences of the United States of America 116: 22657–22663. Crossref
Kawahara, A.Y., Storer, C., Carvalho, A.P.S., Plotkin, D.M., Condamine, F.L., Braga, M.P., Ellis, E.A., St Laurent, R.A., Li, X., Barve, V., Cai, L., Earl, C., Frandsen, P.B., Owens, H.L., Valencia-Montoya, W.A., Aduse-Poku, K., Toussaint, E.F.A., Dexter, K.M., Doleck, T., Markee, A., Messcher, R., Nguyen, Y.-L., Badon, J.A.T., Benítez, H.A., Braby, M.F., Buenavente, P.A.C., Chan, W.-P., Collins, S.C., …{?} and Lohman, D.J. 2023. A global phylogeny of butterflies reveals their evolutionary history, ancestral hosts and biogeographic origins. Nature Ecology and Evolution 7: 903–913. Crossref
Kozlov, M.V., Ivanov, V.D., and Rasnitsyn, A.P. 2002. Order Lepidoptera Linné, 1758. The butterflies and moths (= Papilionida Laicharting, 1781). In: A.P. Rasnitsyn and D.L.J. Quicke (eds.), History of Insects, 220–227. Kluwer Academic Publishers, Dordrecht.
Kristensen, N.P., and Skalski, A.W. 1998. Phylogeny and palaeontology. In: N.P. Kristensen (ed.), Handbook of Zoology. Volume IV. Arthropoda: Insecta. Part 35. Lepidoptera, Moths and Butterflies 1: Evolution, Systematics, and Biogeography, 7–25. W. de Gruyter, Berlin. Crossref
Labandeira, C.C. and Sepkoski, T.J., Jr. 1993. Insect diversity in the fossil record. Science 261: 310–315. Crossref
Lutz, H. 1984. Beitrag zur Kenntnis der Unteroligozänen Insektenfauna von Ceresté (Süd-Frankreich). Documenta naturae 21: 1–25.
Lutz, H. 1985a. Eine fossile Stechmücke aus dem Unter-Oligozän von Céreste, Frankreich (Diptera: Culicidae). Paläontologische Zeitschrift 59: 269–275. Crossref
Lutz, H. 1985b. Eine fossile Stielaugenfliege aus dem Unter-Oligozän von Céreste, Frankreich (Diptera: Culicidae). Paläontologische Zeitschrift 59: 75–78. Crossref
Martínez-Delclòs, X. and Martinell, J. 1993. Insect taphonomy experiments. Their application to the Cretaceous outcrops of lithographic limestones from Spain. Kaupia, Darmstadter Beitrage zur Naturgeschichte 2: 133–144.
Masui, A., Bozano, G.C., and Floriani, A. 2011. Nymphalidae Part IV, Subfamily Apaturinae. In: G.C. Bozano (ed.), Guide to the Butterflies of the Palearctic Region, 1–182. Omnes Artes, Milano.
Mayr, G. 2000. Charadriiform birds from the early Oligocene of Céreste (France) and the Middle Eocene of Messel (Hessen, Germany). Geobios 33: 625–636. Crossref
Nel, A., Wei, M., Niu, G.,
Coster, P., Boderau, M., Josse, H., Kundura, J.-P., Kundura, M.-H.,
Brisac, P., Boudet, L., and Jouault, C. 2024.
The first sawfly from the Oligocene of Céreste (Southern France)
(Hymenoptera: Tenthredinidae). European
Journal of Taxonomy 917: 1–18.
Nidergas, V., Hadrava, J., Garrouste, R., Prokop, J., Schubnel T., and Nel, A. 2018. The first pipizine hoverfly from the Oligocene of Céreste, France. Acta Palaeontologica Polonica 63: 539–548. Crossref
Nieukerken, E.J. van, Kaila, L., Kitching, I.J., Kristensen, N.P., Lees, D.C., Minet, J., Mitter, C., Mutanen, M., Regier, J.C., Simonsen, T.J., Wahlberg, N., Yen, S.-H., Zahiri, R., Adamski, D., Baixeras, J., Bartsch, D., Bengtsson, B.Å., Brown, J.W., Bucheli, S.R., Davis, D.R., De Prins, J., De Prins, W., Epstein, M.E., Gentili-Poole, P., Gielis, C., Hattenschwiler, P., Hausmann, A., Holloway, J.D., Kallies, A., Karsholt, O., Kawahara, A.Y., Koster, J.C., Kozlov, M.V., Lafontaine, J.D., Lamas, G., Landry, J.-F., Lee, S., Nuss, M., Park, K.-T., Penz, C., Rota, J., Schintlmeister, A., Schmidt, B.C., Sohn, J.-C., Solis, M.A., Tarmann, G.M., Warren, A.D., Weller, S., Yakovlev, R.V., Zolotuhin, V.V., and Zwick, A. 2011. Order Lepidoptera Linnaeus, 1758. In: Z.-Q. Zhang (ed.), Animal biodiversity: an outline of higher-level classification and survey of taxonomic richness. Zootaxa 3148: 212–221. Crossref
Ohshima, I., Tanikawa-Dodo, Y., Saigusa, T., Nishiyama, T., Kitani, M., Hasebe, M., and Mohri, H. 2010. Phylogeny, biogeography, and host-plant association in the subfamily Apaturinae (Insecta: Lepidoptera: Nymphalidae) inferred from eight nuclear and seven mitochondrial genes. Molecular Phylogenetics and Evolution 57: 1026–1036. Crossref
Peñalver, E. and Grimaldi, D. 2006. New data on Miocene butterflies in Dominican amber with the description of a new nymphalid. American Museum Novitates 3519: 1–17. Crossref
Pfretzschner, H.-U. 1998. Ein weiteres Exemplar von Lethe? corbieri Nel, 1993 (Lepidoptera, Satyridae) aus dem Unter-Oligozän von Céreste (Süd-Frankreich). Palaeontologische Zeitschrift 72: 59–64. Crossref
Sarto i Monteys, V., Hausmann, A., Solórzano-Kraemer, M.M., Hammel, J.U., Baixeras, J., Delclòs, X., and Peñalver, E. 2022. A new fossil inchworm moth discovered in Miocene Dominican amber (Lepidoptera: Geometridae). Journal of South American Earth Sciences 120 (2022): 104055. Crossref
Sohn, J.-C., Labandeira, C., and Davis, D. 2015. The fossil record and taphonomy of butterflies and moths (Insecta, Lepidoptera): implications for evolutionary diversity and divergence-time estimates. BMC Evolutionary Biology 15 (12): 1–15. Crossref
Sohn, J.-C., Labandeira, C., Davis, D., and Mitter C. 2012. An annotated catalog of fossil and subfossil Lepidoptera (Insecta: Holometabola) of the world. Zootaxa 3286: 1–132. Crossref
Van Eldijk, T.J., Wappler, T., Strother, P.K., van der Weijst, C.M., Rajaei, H., van de Visscher, H., and Schootbrugge, B.J.S. 2018. A Triassic–Jurassic window into the evolution of Lepidoptera. Science Advances 4 (1): e1701568. Crossref
Wahlberg, N., Leneveu, J., Kodandaramaiah, U., Peña, C., Nylin, S., Freitas, A.V.L., and Brower, A.V.Z. 2009. Nymphalid butterflies diversify following near demise at the Cretaceous/Tertiary boundary. Proceedings of the Royal Society Series B Biological Sciences 276: 4295–4302. Crossref
Wahlberg, N., Wheat, C.W., and Peña, C. 2013. Timing and patterns in the taxonomic diversification of Lepidoptera (butterflies and moths). PLOS ONE 8 (11): e80875. Crossref
Wappler, T. 2004. The first lace bug (Insecta: Heteroptera: Tingidae) from the Lower Oligocene deposits near Céreste, France. Neues Jahrbuch für Geologie und Paläontologie, Monatshefte 2004 (5): 278–288. Crossref
Whalley, P.E.S. 1985. The systematics and palaeogeography of the Lower Jurassic insects of Dorset, England. Bulletin of the British Museum (Natural History), Geology 39 (3): 107–189.
Acta Palaeontol. Pol. 71 (1): 185–191, 2026
http://doi.org/10.4202/app.01332.2026